📊 Annual Product Quality Review (APQR): Complete Guide with WHO GMP Requirements, Compliance Strategy & Industry Practice
🔍 Introduction
In the pharmaceutical industry, ensuring consistent product quality is essential for patient safety, regulatory compliance, and global market access. One of the most critical systems used to achieve this is the Annual Product Quality Review (APQR).
In Bangladesh, where pharmaceutical companies are increasingly exporting to regulated markets, APQR is a mandatory quality system aligned with international standards such as WHO-GMP, EU-GMP, and ICH guidelines.
👉 Visit https://pharmajobaid.com/ for more pharma insights and career resources.

📘 What is APQR?
Annual Product Quality Review (APQR) is a systematic, documented evaluation of a pharmaceutical product conducted annually to confirm:
- Consistency of manufacturing processes
- Compliance with specifications
- Identification of trends
- Need for improvements (CAPA)
Other terms:
- PQR (Product Quality Review) – EU
- APR (Annual Product Review) – US FDA
🌍 WHO GMP Requirements for APQR (CRITICAL SECTION)
The World Health Organization (WHO) clearly defines APQR requirements under:
👉 WHO Technical Report Series (TRS) 986, Annex 2
👉 Updated in WHO TRS 1025 & later revisions
📜 Key WHO Requirements for APQR
According to WHO GMP, manufacturers must conduct regular (at least annual) product quality reviews to verify consistency and compliance.
✅ WHO-Mandated Elements of APQR:
- Review of Starting Materials
- Quality of APIs and excipients
- Supplier performance
- Critical In-Process Controls
- Process parameters
- Environmental monitoring
- Finished Product Results
- Batch release data
- Specification compliance
- Deviations & Non-Conformances
- All significant deviations
- Investigation outcomes
- OOS (Out of Specification) Results
- Trends and root causes
- Stability Data Review
- Ongoing stability program
- Shelf-life verification
- Quality Complaints & Recalls
- Market complaints
- Adverse trends
- CAPA Review
- Effectiveness of corrective actions
- Process & Analytical Changes
- Change control impact
- Validation Status
- Process validation
- Cleaning validation
- Qualification Status
- Equipment & utilities
- Contractual Agreements (if applicable)
- Contract manufacturing oversight
⚠️ WHO Expectation (Important Insight)
WHO emphasizes that APQR should:
- ✔ Identify trends over time
- ✔ Highlight process variability
- ✔ Detect recurrent issues
- ✔ Support continuous improvement
- ✔ Be reviewed by senior management
👉 Failure to perform proper APQR can lead to WHO GMP non-compliance observations during audits.
🇧🇩 APQR in Bangladesh (DGDA Perspective)
The Directorate General of Drug Administration (DGDA) aligns closely with WHO GMP and expects:
- Annual APQR reports for each product
- Availability during inspections
- Evidence of CAPA and follow-up actions
Bangladeshi export-oriented companies must strictly follow WHO APQR requirements to maintain:
- Export licenses
- International approvals
- Market credibility
🎯 Objectives of APQR
- Ensure product quality consistency
- Detect trends and risks
- Improve manufacturing processes
- Strengthen GMP compliance
- Support regulatory inspections
🔄 APQR Lifecycle
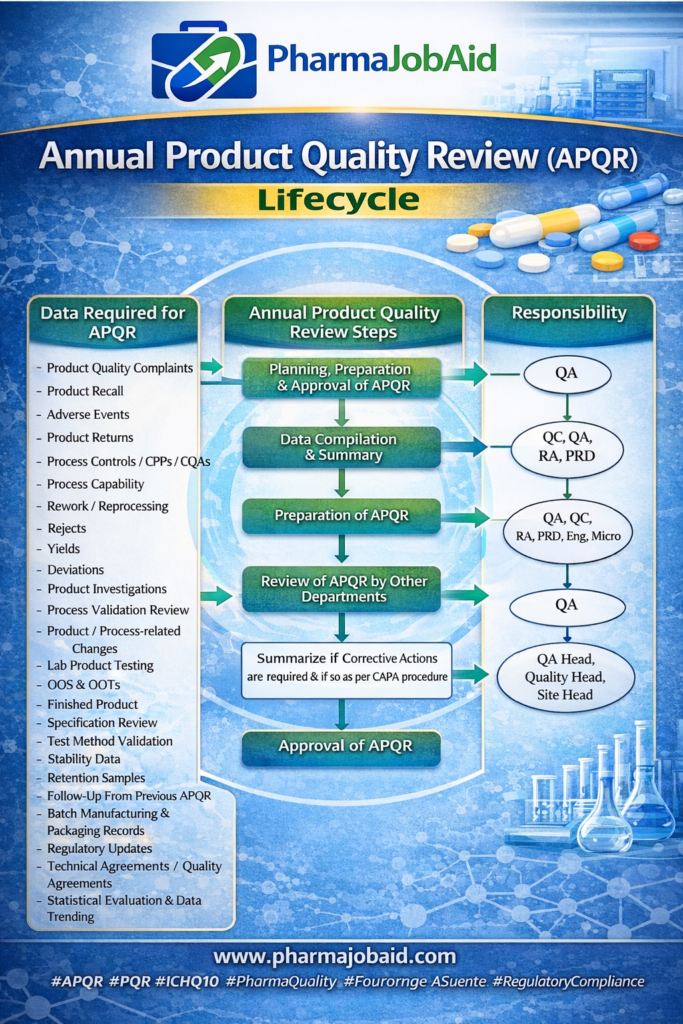
Steps:
- Data Collection
- Data Analysis
- Trend Evaluation
- CAPA Identification
- Report Preparation
- QA Approval & Management Review
📊 Key Elements of APQR (WHO-Aligned)
- Batch production review
- Deviations & OOS
- Stability data
- Complaints & recalls
- Change control
- Validation status
- CAPA effectiveness
📉 Common APQR Findings in Bangladesh
- Increasing deviation trends
- Recurrent OOS issues
- Stability failures
- Weak CAPA implementation
- Documentation gaps
⚙️ Practical Example (WHO-Aligned APQR)
Product: Paracetamol Tablets (Bangladesh facility)
Issue:
- Increasing dissolution failures observed
WHO APQR Review Outcome:
- Trend confirmed over multiple batches
- Linked to raw material variability
CAPA:
- Supplier requalification
- Tightened specifications
- Process optimization
Result:
- Improved batch consistency
- Reduced regulatory risk
🧠 Challenges in Bangladesh Pharma
- Manual data handling
- Lack of statistical tools
- Limited GMP training
- Weak data integrity culture
🚀 Best Practices (WHO-Compliant)
- Use electronic QMS
- Apply statistical trend analysis
- Integrate risk management (ICH Q9)
- Ensure data integrity (ALCOA+)
- Conduct periodic internal audits
- Train teams on WHO GMP
📈 Importance for Export Compliance
WHO-compliant APQR is essential for:
- WHO prequalification
- EU-GMP inspections
- International tenders
- Regulatory approvals
🔗 Internal Linking (PharmaJobAid SEO)
- QA in Pharma
- CAPA Management
- Deviation Handling
- Validation & Qualification
🌐 External Backlinks
- WHO GMP Guidelines
- US FDA (21 CFR 211.180)
- ICH Q10 Pharmaceutical Quality System
- EMA GMP Guidelines
🎯 Conclusion
The WHO-mandated APQR system is a cornerstone of pharmaceutical quality management. For Bangladesh’s growing pharma industry, implementing a robust APQR process ensures:
- Regulatory compliance
- Product consistency
- Global market competitiveness
For professionals, mastering WHO APQR requirements opens strong career pathways in QA, Regulatory Affairs, and Compliance.
