CAPA in the Pharmaceutical Industry in Bangladesh: Complete Guide to Corrective and Preventive Action (GMP, SOPs, Real Cases & Career Insights)
Focus Keywords:
CAPA in pharmaceutical industry, corrective and preventive action pharma, CAPA Bangladesh pharma, GMP CAPA system, deviation CAPA pharma, quality assurance pharma Bangladesh
📌 Introduction
In the highly regulated pharmaceutical industry of Bangladesh, maintaining product quality, patient safety, and regulatory compliance is non-negotiable. One of the most critical systems ensuring this is CAPA (Corrective and Preventive Action).
CAPA is not just a regulatory requirement—it is the backbone of continuous improvement in pharmaceutical manufacturing, quality assurance, and compliance management.
Bangladesh’s pharmaceutical sector, regulated by the Directorate General of Drug Administration (DGDA) and aligned with global standards like WHO GMP, US FDA, and EMA, places strong emphasis on CAPA systems.
👉 For more pharma career insights, visit: https://pharmajobaid.com/
🧪 What is CAPA in Pharmaceutical Industry?
CAPA stands for:
- Corrective Action → Fixing an existing problem
- Preventive Action → Preventing recurrence or future issues
📖 Definition (GMP Perspective)
CAPA is a systematic process used to:
- Identify deviations or non-conformances
- Investigate root causes
- Implement solutions
- Prevent recurrence
🔍 Why CAPA is Critical in the Bangladesh Pharma Industry
Bangladesh exports pharmaceuticals to over 150 countries. To maintain this global trust, CAPA ensures:
✅ Key Benefits
- Compliance with WHO GMP guidelines
- Improved product quality and safety
- Reduction of batch failures
- Strong audit readiness
- Continuous process improvement
📊 Industry Reality
Top companies like:
- Beximco Pharma
- Square Pharmaceuticals
- Incepta Pharmaceuticals
Maintain strict CAPA systems to meet international regulatory standards.
🏭 Sources of CAPA in Pharma
CAPA can originate from multiple quality events:
📌 Common CAPA Triggers
- Deviation
- Out of Specification (OOS)
- Out of Trend (OOT)
- Customer complaints
- Audit observations
- Product recalls
- Equipment failures
👉 Read more: Internal link suggestion
➡️ Deviation in Pharmaceutical Industry in Bangladesh (PharmaJobAid)
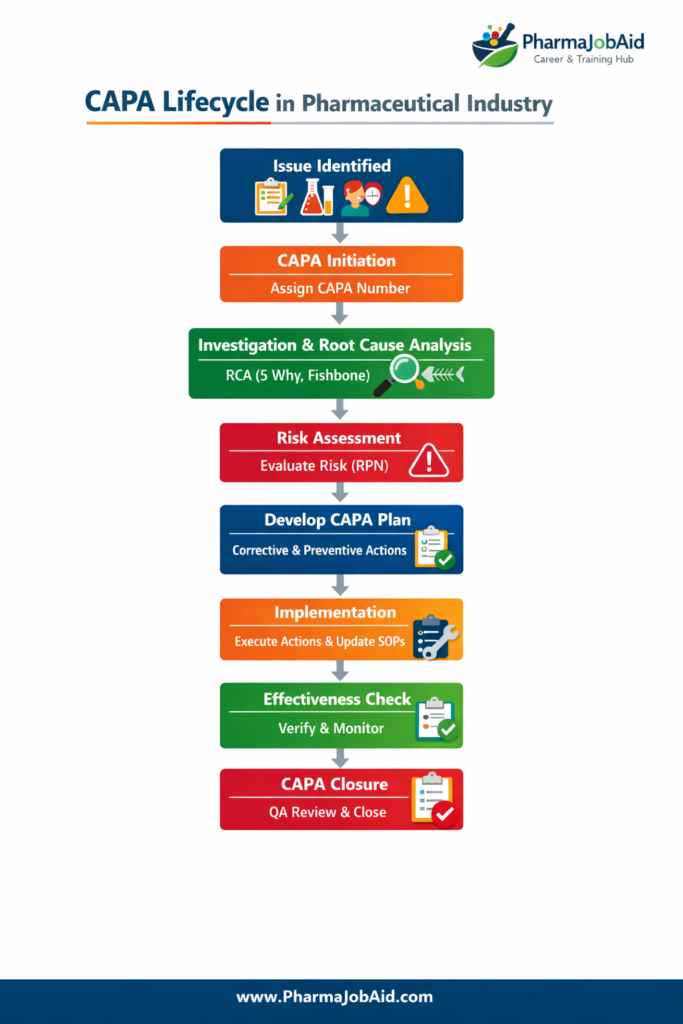
🔄 CAPA Lifecycle in Pharmaceutical Industry (Detailed GMP Guide for Bangladesh)
The Corrective and Preventive Action (CAPA) lifecycle is a structured, step-by-step system used in pharmaceutical companies to investigate issues, eliminate root causes, and prevent recurrence.
In Bangladesh, CAPA systems are aligned with global standards such as World Health Organization GMP, U.S. Food and Drug Administration, and enforced locally by the Directorate General of Drug Administration (DGDA).
🧭 Overview of CAPA Lifecycle
A complete CAPA lifecycle includes 7 critical stages:
- Identification
- CAPA Initiation
- Investigation & Root Cause Analysis
- Risk Assessment
- CAPA Plan Development
- Implementation
- Effectiveness Check & Closure
1️⃣ Identification of Issue (Problem Detection)
📌 What Happens Here?
The CAPA process begins when a quality issue is identified.
🔍 Sources of Identification
- Deviation reports
- OOS (Out of Specification) results
- OOT (Out of Trend)
- Customer complaints
- Internal or external audits
- Equipment breakdown
- Stability failures
🧪 Example (Bangladesh Industry)
During tablet compression, hardness falls below limit → Deviation raised → CAPA required
⚠️ Key Requirement
- Immediate documentation
- No delay in reporting
- Follow Good Documentation Practices (GDP/GDocP)
2️⃣ CAPA Initiation
📌 What Happens Here?
- CAPA record is officially opened
- Unique CAPA number assigned
📄 Typical Fields
- CAPA ID (e.g., QA/CAPA/2026/015)
- Source (Deviation/OOS/Audit)
- Department
- Date of initiation
👨⚕️ Responsible Department
Usually initiated by:
- QA (Quality Assurance)
- QC
- Production
🧠 Pro Tip
In top Bangladeshi pharma companies, CAPA initiation is done through eQMS systems for traceability.
3️⃣ Investigation & Root Cause Analysis (RCA)
📌 Objective
To identify the true root cause, not just symptoms.
🔍 Investigation Activities
- Batch record review
- Equipment logbook review
- Interview operators
- Environmental condition check
- Material analysis
🧠 Root Cause Analysis Tools
✔️ 5 Why Analysis
Example:
- Why issue occurred? → Machine error
- Why machine error? → Calibration missed
- Why missed? → No schedule
👉 Root Cause = Lack of preventive maintenance system
✔️ Fishbone Diagram (Ishikawa)
Categories:
- Man
- Machine
- Method
- Material
- Environment
⚠️ Common Mistake
❌ Blaming operator without systemic investigation
✅ Focus on process/system failure
4️⃣ Risk Assessment
📌 Why It Matters?
Not all issues have the same impact. Risk assessment helps prioritize CAPA.
📊 Risk Parameters
- Severity (Impact on patient/product)
- Occurrence (Frequency)
- Detectability
🧮 Risk Priority Number (RPN)
RPN = Severity × Occurrence × Detectability
🧪 Example
| Factor | Score |
|---|---|
| Severity | 9 |
| Occurrence | 4 |
| Detectability | 3 |
👉 RPN = 108 → High risk → Immediate CAPA
📌 Regulatory Expectation
Risk-based CAPA is required by:
- WHO GMP
- FDA
- EU GMP
5️⃣ CAPA Plan Development
📌 What Happens Here?
A structured action plan is created.
📄 Components of CAPA Plan
🔧 Corrective Actions (Short-term)
- Fix immediate issue
- Example: Recalibrate machine
🔮 Preventive Actions (Long-term)
- Prevent recurrence
- Example: Monthly calibration schedule
📋 Plan Must Include
- Action description
- Responsible person
- Timeline
- Required resources
- SOP updates (if needed)
⚠️ Key Principle
Corrective ≠ Preventive
Many companies incorrectly write same action for both.
6️⃣ CAPA Implementation
📌 Execution Phase
All planned actions are implemented.
🔧 Activities
- SOP revision
- Training of personnel
- Equipment maintenance
- Process modification
📊 Documentation Required
- Training records
- Calibration reports
- Updated SOP copies
- Implementation evidence
⚠️ Common Failure
❌ CAPA written but not implemented
✅ Evidence-based execution required
7️⃣ Effectiveness Check & Verification
📌 Purpose
To confirm CAPA actually worked.
🔍 Methods
- Monitor next 3–5 batches
- Trend analysis
- Repeat testing
- Audit review
🧪 Example
After calibration:
- No deviation in next batches → CAPA effective
⚠️ Mistake
❌ Closing CAPA without verification
✅ Effectiveness check mandatory
8️⃣ CAPA Closure
📌 Final Step
CAPA is closed only after:
- All actions completed
- Effectiveness verified
- QA approval
📄 Closure Documentation
- CAPA summary
- Evidence attached
- QA sign-off
- Closure date
👨⚕️ Authority
Final closure is done by:
- QA Manager / Head QA
🔁 CAPA Lifecycle Flow Summary
Identification → Initiation → Investigation → RCA → Risk Assessment
→ CAPA Plan → Implementation → Effectiveness Check → Closure
🇧🇩 Real Industry Insight (Bangladesh)
In leading companies like:
- Square Pharma
- Beximco Pharma
- Incepta
CAPA systems are:
- Digitally tracked
- Integrated with deviation & change control
- Reviewed during audits
🚀 Advanced CAPA Practices (Pharma 4.0)
Modern CAPA systems include:
- eQMS software
- Automated alerts
- KPI dashboards
- Trend analytics
📊 CAPA KPIs (Key Performance Indicators)
- CAPA closure time
- Recurrence rate
- Overdue CAPA (%)
- Effectiveness success rate
❌ Common CAPA Failures in Bangladesh Pharma
- Weak root cause analysis
- Delayed CAPA closure
- Lack of training
- Poor documentation
- No trend analysis
✅ Best Practices
- Strong QA oversight
- Risk-based approach
- Real root cause identification
- Continuous monitoring
- Proper training
📌 Conclusion
A robust CAPA lifecycle ensures:
- GMP compliance
- Product quality
- Patient safety
- Audit success
For Bangladesh’s growing pharmaceutical export market, CAPA is not optional—it is essential.
📢 Want More?
I can create next:
✅ CAPA Flowchart infographic (PharmaJobAid branded PNG)
✅ Real CAPA investigation form (editable template)
✅ Case study (FDA audit CAPA failure example)
✅ Elementor visual layout for this section
Just tell me 👍
📊 Example of CAPA in Bangladesh Pharma Industry
🧪 Real Case: Tablet Weight Variation
Problem:
Tablet weight variation out of limit during compression
Root Cause:
Improper machine calibration
Corrective Action:
- Recalibrate machine
- Retrain operator
Preventive Action:
- Schedule periodic calibration
- Update SOP
Effectiveness Check:
No deviation observed in next 3 batches
⚙️ CAPA vs Deviation vs Change Control
| System | Purpose |
|---|---|
| Deviation | Identify problem |
| CAPA | Fix & prevent problem |
| Change Control | Manage changes |
👉 Internal Link Suggestion:
➡️ Change Control in Pharmaceutical Industry
🧠 Root Cause Analysis Tools Explained
🔍 5 Why Analysis Example
Why deviation occurred?
→ Machine not calibrated
Why not calibrated?
→ Maintenance missed
Why missed?
→ No schedule
👉 Root Cause: Lack of preventive maintenance system
🐟 Fishbone Diagram Categories
- Man
- Machine
- Method
- Material
- Environment
📏 CAPA and Regulatory Compliance
CAPA is mandatory under:
- WHO GMP
- US FDA (21 CFR Part 211)
- EU GMP
- PIC/S Guidelines
📌 External Reference Links
- WHO GMP Guidelines: https://www.who.int
- US FDA CAPA Guidance: https://www.fda.gov
- EMA Quality Guidelines: https://www.ema.europa.eu
🇧🇩 CAPA in Bangladesh Regulatory Perspective
In Bangladesh:
- DGDA audits require CAPA documentation
- CAPA linked with:
- Validation
- Qualification
- Risk management
CAPA is a major focus during:
- WHO inspections
- Export audits
- Customer audits
📉 Common CAPA Failures in Pharma Industry
❌ Mistakes to Avoid
- Weak root cause analysis
- Copy-paste CAPA
- No effectiveness check
- Delayed closure
- Lack of QA oversight
🚀 Best Practices for Effective CAPA System
✅ Industry Best Practices
- Use risk-based CAPA
- Train employees regularly
- Digital CAPA tracking systems
- Strong QA review
- Trend analysis
💻 Digital CAPA Systems (Pharma 4.0)
Modern companies in Bangladesh are adopting:
- Electronic QMS (eQMS)
- Automated CAPA tracking
- Data analytics
👉 Read more:
➡️ Pharma 4.0 in Bangladesh (PharmaJobAid)
👨⚕️ Career Opportunities in CAPA (Bangladesh Pharma)
CAPA is a core skill for:
🎯 Job Roles
- QA Officer
- QA Executive
- Compliance Specialist
- Validation Engineer
💼 Required Skills
- GMP knowledge
- Investigation skills
- Documentation
- Analytical thinking
