Deviation in Pharmaceutical Industry in Bangladesh: Advanced GMP Guide for Investigation, Compliance & Quality Excellence
Introduction
The pharmaceutical industry in Bangladesh has transformed into a globally recognized sector, supplying approximately 97–98% of domestic demand and exporting medicines to over 150 countries. With this expansion, regulatory expectations have intensified, particularly in areas of quality assurance, compliance, and documentation control.
One of the most critical pillars of pharmaceutical quality systems is deviation management—a structured process to identify, document, investigate, and resolve unexpected events that may impact product quality or compliance.
Deviation is not just a regulatory checkbox. It is a core component of Quality Management System (QMS) that ensures:
- Patient safety
- Product integrity
- Regulatory compliance
- Continuous improvement
In Bangladesh, deviation handling is governed by WHO-GMP, DGDA regulations, and increasingly by international standards such as US FDA, EMA, and ICH guidelines.
👉 For pharma QA training, SOP guides, and job opportunities, visit:
https://pharmajobaid.com/
What is Deviation in Pharmaceutical Industry?
A deviation refers to any departure from approved processes, procedures, specifications, or established standards.
According to GMP frameworks:
A deviation is any unplanned event that diverges from approved instructions or processes and has the potential to affect product quality, safety, or regulatory compliance.
📌 External Reference:
WHO GMP Guidelines – https://www.who.int/publications/i/item/9789241549950
Key Characteristics of Deviation
- Unexpected or unplanned event
- Occurs during manufacturing, testing, packaging, or storage
- May or may not impact product quality
- Requires documentation, investigation, and closure
Deviation can occur at any stage of the pharmaceutical lifecycle, including:
- Raw material handling
- Manufacturing process
- Quality control testing
- Packaging operations
- Warehouse storage
Why Deviation Management is Critical in Pharma
Deviation management is essential because pharmaceutical products directly affect human health. Even minor process variations can lead to serious consequences.
1. Ensuring Product Quality
Deviation systems ensure that all processes remain within validated parameters. Any variation is immediately investigated to prevent compromised product quality.
2. Protecting Patient Safety
Uncontrolled deviations can result in:
- Substandard drugs
- Contamination
- Incorrect dosage
Proper deviation handling prevents such risks.
3. Regulatory Compliance
Regulatory agencies require strict deviation control:
- US FDA – https://www.fda.gov/drugs/pharmaceutical-quality-resources
- EMA – https://www.ema.europa.eu
- ICH Q10 – https://www.ich.org/page/quality-guidelines
Failure to manage deviations can lead to:
- Warning letters
- Import bans
- License suspension
4. Continuous Improvement
Deviation data helps identify:
- Process weaknesses
- Training gaps
- System inefficiencies
Types of Deviation in Pharmaceutical Industry
1. Critical Deviation
These have a direct impact on product quality and patient safety.
Examples:
- Sterility failure in injectable products
- Cross-contamination between batches
- Labeling mix-up
Impact:
- Product recall
- Regulatory action
- Serious patient risk
2. Major Deviation
Moderate impact on product quality or compliance.
Examples:
- Equipment malfunction
- Process parameter deviation
- Incomplete batch documentation
3. Minor Deviation
Minimal or no impact on product quality.
Examples:
- Documentation errors
- Minor environmental fluctuations
👉 These classifications align with real GMP industry practices
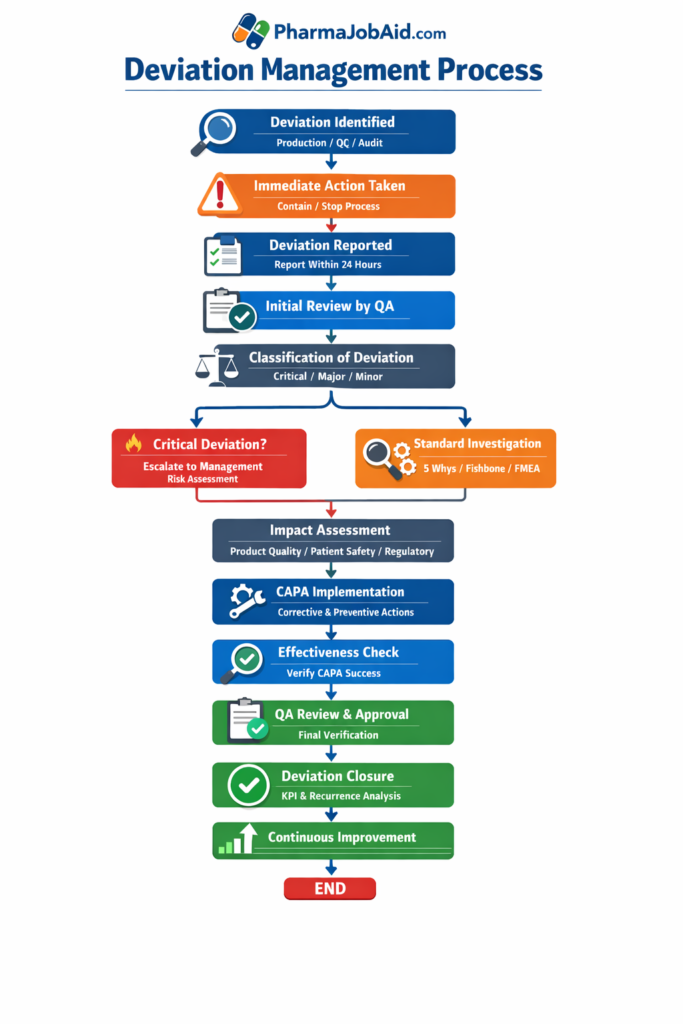
📊 Deviation Management Flowchart (Pharmaceutical Industry)
START
↓
Deviation Identified
(Production / QC / IPQA / Audit)
↓
Immediate Action Taken
(Containment / Stop process if needed)
↓
Deviation Reported
(Fill Deviation Form within 24 hours)
↓
Initial Review by QA
↓
Classification of Deviation
(Critical / Major / Minor)
↓
Is Critical?
├── YES → Immediate Escalation to QA Head / Management
│ ↓
│ Risk Assessment (High Priority)
│
└── NO → Continue Standard Investigation
↓
Investigation Initiated
(Cross-functional team: QA + QC + Production)
↓
Root Cause Analysis (RCA)
(5 Whys / Fishbone / FMEA)
↓
Identify Root Cause
↓
Impact Assessment
(Product Quality / Patient Safety / Regulatory)
↓
Is Product Affected?
├── YES → Batch Rejection / Recall / Reprocessing
└── NO → Continue
↓
CAPA Plan Development
(Corrective + Preventive Actions)
↓
CAPA Implementation
↓
Effectiveness Check
(Verification after defined period)
↓
QA Review & Approval
↓
Deviation Closure
↓
Trending & Monitoring
(KPI / Recurrence analysis)
↓
Continuous Improvement
↓
END
Classification Based on Occurrence
Planned Deviation
A pre-approved, controlled deviation from an SOP.
Example:
- Using alternate equipment during maintenance
Unplanned Deviation
Unexpected event requiring immediate action.
Example:
- Temperature excursion during manufacturing
👉 Widely practiced in Bangladesh pharma industries
Root Causes of Deviation (Detailed Analysis)
Understanding root causes is the foundation of effective CAPA.
1. Human Error
Most common cause in pharma.
Reasons:
- Lack of training
- SOP misinterpretation
- Fatigue or negligence
2. Equipment Failure
Causes:
- Improper maintenance
- Calibration issues
- Mechanical breakdown
3. Material Issues
Causes:
- Supplier inconsistency
- Raw material contamination
4. Environmental Factors
Examples:
- Temperature excursions
- Humidity variation
5. Process Failures
Examples:
- Incorrect mixing time
- Parameter deviation
6. Documentation Errors
Examples:
- Missing signatures
- Incorrect entries
👉 These causes are widely documented in industry scenarios
Deviation Lifecycle (Industry-Level Explanation)
A structured deviation lifecycle ensures traceability and compliance.
Step 1: Identification
Detected through:
- Production monitoring
- QC testing
- Internal audits
- IPQA
Step 2: Immediate Reporting
Deviation must be reported within 24 hours.
Step 3: Classification
Categorized into:
- Critical
- Major
- Minor
Step 4: Investigation
Root cause analysis performed using structured tools.
Step 5: Impact Assessment
Evaluate:
- Product quality
- Patient safety
- Regulatory risk
Step 6: CAPA Implementation
Corrective and preventive actions implemented.
Step 7: Closure
QA verifies and approves closure.
👉 This lifecycle reflects real GMP workflow
Advanced Investigation Tools (Deep Dive)
1. 5 Whys Analysis
Simple yet powerful.
2. Fishbone Diagram
Categorizes causes into:
- Man
- Machine
- Method
- Material
- Environment
3. FMEA (Failure Mode and Effects Analysis)
Risk-based approach prioritizing high-risk deviations.
📌 ICH Q9:
https://www.ich.org/page/quality-guidelines
CAPA (Corrective and Preventive Action)
CAPA ensures long-term system improvement.
Corrective Action
Fix current issue.
Preventive Action
Prevent recurrence.
Deviation vs OOS vs OOT (Advanced Comparison)
| Parameter | Deviation | OOS | OOT |
|---|---|---|---|
| Nature | Process issue | Test result failure | Trend shift |
| Trigger | Process variation | Lab result | Statistical trend |
| Action | Investigation + CAPA | Lab investigation | Trend analysis |
📌 FDA OOS Guidance:
https://www.fda.gov/media/71001/download
Role of Quality Assurance (QA)
QA is the central authority in deviation management.
Responsibilities:
- Review deviation reports
- Approve investigations
- Ensure timely closure
- Monitor CAPA effectiveness
- Ensure data integrity
QA ensures compliance with GMP and regulatory standards.
Deviation Management in Bangladesh Pharma Industry
Bangladesh operates under DGDA regulations aligned with WHO-GMP.
📌 DGDA:
https://dgda.gov.bd
Industry Practices
- Strong SOP-based systems
- Electronic QMS platforms
- Frequent audits
- Export compliance
Leading Companies
- Square Pharmaceuticals
- Beximco Pharma
- Incepta Pharmaceuticals
These companies follow global compliance frameworks.
Regulatory Expectations (Advanced Insight)
WHO GMP
Requires full deviation documentation.
US FDA
Focus on investigation and CAPA.
EU GMP
Strong emphasis on data integrity.
ICH Q10
Defines pharmaceutical quality system.
📌 ICH Q10:
https://database.ich.org/sites/default/files/Q10_Guideline.pdf
Real-Life Deviation Examples
Manufacturing
- Temperature excursion
- Mixing error
QC Laboratory
- OOS result
- Instrument failure
Warehouse
- Improper storage
Documentation
- Missing signatures
👉 Common real-world examples
Consequences of Poor Deviation Management
- Product recall
- Regulatory warning letters
- Export restrictions
- Financial loss
- Brand damage
Best Practices (Advanced GMP Level)
1. Strong Quality Culture
Encourage open reporting.
2. Data Integrity (ALCOA+)
Ensure reliable documentation.
3. Cross-Functional Teams
QA, QC, Production collaboration.
4. Continuous Training
Regular GMP updates.
5. Trend Analysis
Identify recurring issues.
Deviation Trending & Analytics
Helps identify:
- Repeated failures
- Process weaknesses
Tools:
- KPI dashboards
- Statistical analysis
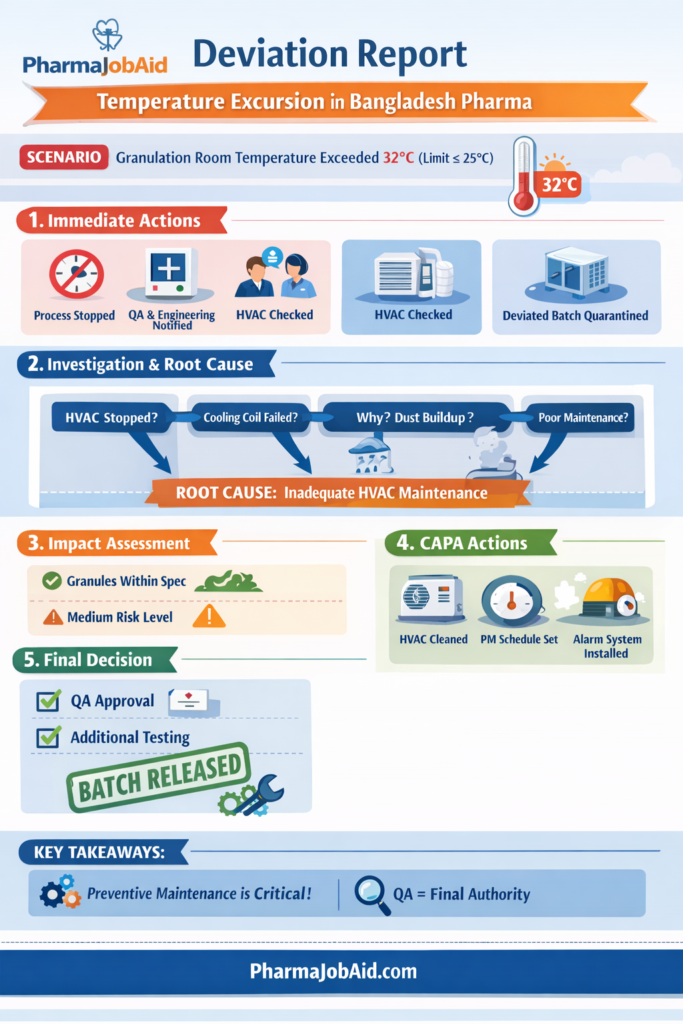
📄 Deviation Report Form (Real Industry Example – Bangladesh Pharma)
🔷 Section 1: Basic Information
Deviation No: DEV/2026/QA/045
Date of Occurrence: 15 March 2026
Date of Reporting: 15 March 2026
Department: Production – Tablet Section
Product Name: Paracetamol 500 mg Tablet
Batch No: PCM230315
Stage: Granulation
Reported By: Production Officer
🔷 Section 2: Description of Deviation
During wet granulation, the room temperature exceeded the specified limit (≤25°C) and reached 32°C for approximately 45 minutes due to HVAC malfunction.
The deviation was observed during routine environmental monitoring.
🔷 Section 3: Immediate Action Taken
- Granulation process stopped immediately
- Batch status set to “Under Deviation”
- QA and Engineering notified
- HVAC system inspected
- Temperature restored within limit
🔷 Section 4: Classification of Deviation
- ☐ Minor
- ☑ Major
- ☐ Critical
Justification:
Temperature excursion may impact product quality (moisture-sensitive process) but no direct evidence of product failure.
🔷 Section 5: Investigation Team
- QA Officer (Lead)
- Production Manager
- Engineering Executive
🔷 Section 6: Investigation Details
📌 6.1 What Happened?
Room temperature exceeded validated range during granulation.
📌 6.2 When?
15 March 2026, 10:15 AM – 11:00 AM
📌 6.3 Where?
Granulation Room – Block B
📌 6.4 Who Was Involved?
Production operator and HVAC technician
📌 6.5 How Was It Detected?
Environmental monitoring log + digital sensor alarm
🔷 Section 7: Root Cause Analysis (5 Whys)
- Why temperature increased?
→ HVAC stopped functioning - Why HVAC stopped?
→ Cooling coil malfunction - Why coil malfunctioned?
→ Dust accumulation - Why dust accumulated?
→ Inadequate preventive maintenance - Why maintenance inadequate?
→ No strict PM schedule enforcement
✅ Root Cause Identified:
Inadequate preventive maintenance of HVAC system
🔷 Section 8: Impact Assessment
Product Quality:
- No visible change in granules
- Process resumed after control
- Within acceptable hold time
Risk Level:
- Medium
Conclusion:
Batch can be conditionally acceptable, subject to additional testing
🔷 Section 9: Corrective Action (CA)
- Clean HVAC cooling coil
- Resume normal operation
- Perform additional in-process checks
🔷 Section 10: Preventive Action (PA)
- Implement monthly HVAC preventive maintenance schedule
- Install alarm system for temperature excursion
- Conduct training for engineering team
🔷 Section 11: CAPA Implementation Responsibility
| Action | Responsible | Timeline |
|---|---|---|
| HVAC cleaning | Engineering | Immediate |
| PM schedule | Engineering Head | 7 days |
| Training | QA | 14 days |
🔷 Section 12: Effectiveness Check
After 30 days:
- No repeat deviation observed
- HVAC functioning properly
✅ CAPA found effective
🔷 Section 13: QA Review & Approval
Reviewed By: QA Manager
Approved By: Head of Quality
Final Decision:
✔ Batch Released
🔷 Section 14: Deviation Closure
Closure Date: 20 April 2026
Status: Closed
📊 Key Learning Points (Industry Insight)
- Temperature deviations are very common in Bangladesh pharma plants
- HVAC is a critical GMP system
- Preventive maintenance failures = major root cause
- QA always ensures scientific justification before batch release
🧠 Interview Tip (Very Important)
If asked in interview:
👉 “How do you handle a deviation?”
Answer like this:
- Identify and report immediately
- Take immediate containment action
- Investigate root cause (5 Whys / Fishbone)
- Perform impact assessment
- Implement CAPA
- Verify effectiveness
- QA approval & closure
Digital Transformation in Deviation
Modern systems include:
- eQMS
- AI-based RCA
- Real-time monitoring
Career Opportunities in Deviation
QA
Deviation investigation
QC
Lab deviation handling
Production
Process monitoring
Regulatory Affairs
Compliance documentation
👉 Explore jobs:
https://pharmajobaid.com/
Skills Required
- GMP knowledge
- Analytical thinking
- Documentation skills
- Problem-solving
Future of Deviation Management
- AI-driven systems
- Predictive analytics
- Pharma 4.0 integration
- Global compliance
Conclusion
Deviation management is the backbone of pharmaceutical quality systems. It ensures compliance, protects patients, and drives continuous improvement.
As Bangladesh continues to grow as a global pharmaceutical hub, strong deviation systems will be essential for maintaining quality and regulatory trust.
