Quality Management System (QMS) in the Pharmaceutical Industry in Bangladesh: Complete Guide for Compliance, GMP & Career Growth
Introduction
The pharmaceutical industry in Bangladesh has emerged as one of the most resilient and fast-growing sectors in South Asia. With over 97% of domestic demand met locally and exports reaching more than 160 countries, maintaining world-class quality standards is no longer optional—it is mandatory.
At the heart of this transformation lies the Quality Management System (QMS).
A well-implemented QMS ensures that pharmaceutical companies consistently produce safe, effective, and high-quality medicines while complying with international regulatory standards such as:
- World Health Organization (WHO GMP)
- U.S. Food and Drug Administration (US FDA)
- European Medicines Agency (EU GMP)
- International Council for Harmonisation (ICH Q10, Q9, Q8)
For professionals, understanding QMS is not just about compliance—it is a career accelerator in QA, QC, Regulatory Affairs, and Production.
👉 Explore pharma careers and resources at PharmaJobAid.com
What is a Quality Management System (QMS) in Pharma?
A Quality Management System (QMS) in pharmaceuticals is a structured framework of:
- Policies
- Procedures
- Processes
- Documentation
- Responsibilities
…designed to ensure that pharmaceutical products meet predefined quality standards.
Core Objectives of QMS
- Ensure product safety and efficacy
- Maintain data integrity
- Achieve regulatory compliance
- Promote continuous improvement
- Minimize risk and deviations
Why QMS is Critical in Bangladesh’s Pharmaceutical Industry
Bangladesh’s pharma industry operates under strict regulatory control from:
- Directorate General of Drug Administration (DGDA)
- WHO GMP requirements
- Export compliance (US FDA, EMA, MHRA)
Key Reasons QMS is Essential
- Export Competitiveness
- Required for regulated markets (USA, EU)
- Patient Safety
- Prevents substandard or counterfeit drugs
- Regulatory Approval
- Mandatory for product registration
- Operational Efficiency
- Reduces batch failures and recalls
- Brand Reputation
- Builds trust in global markets
Key Components of Pharmaceutical QMS
A pharmaceutical QMS consists of several interconnected systems:
1. Document Management System


Documentation is the backbone of QMS.
Includes:
- SOPs (Standard Operating Procedures)
- Batch Manufacturing Records (BMR)
- Batch Packaging Records (BPR)
- Logbooks
- Validation documents
Principles:
- ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate)
2. Deviation Management


4
Deviation refers to any departure from approved procedures.
Types:
- Minor deviation
- Major deviation
- Critical deviation
Process:
- Identification
- Investigation
- Root Cause Analysis (RCA)
- CAPA implementation
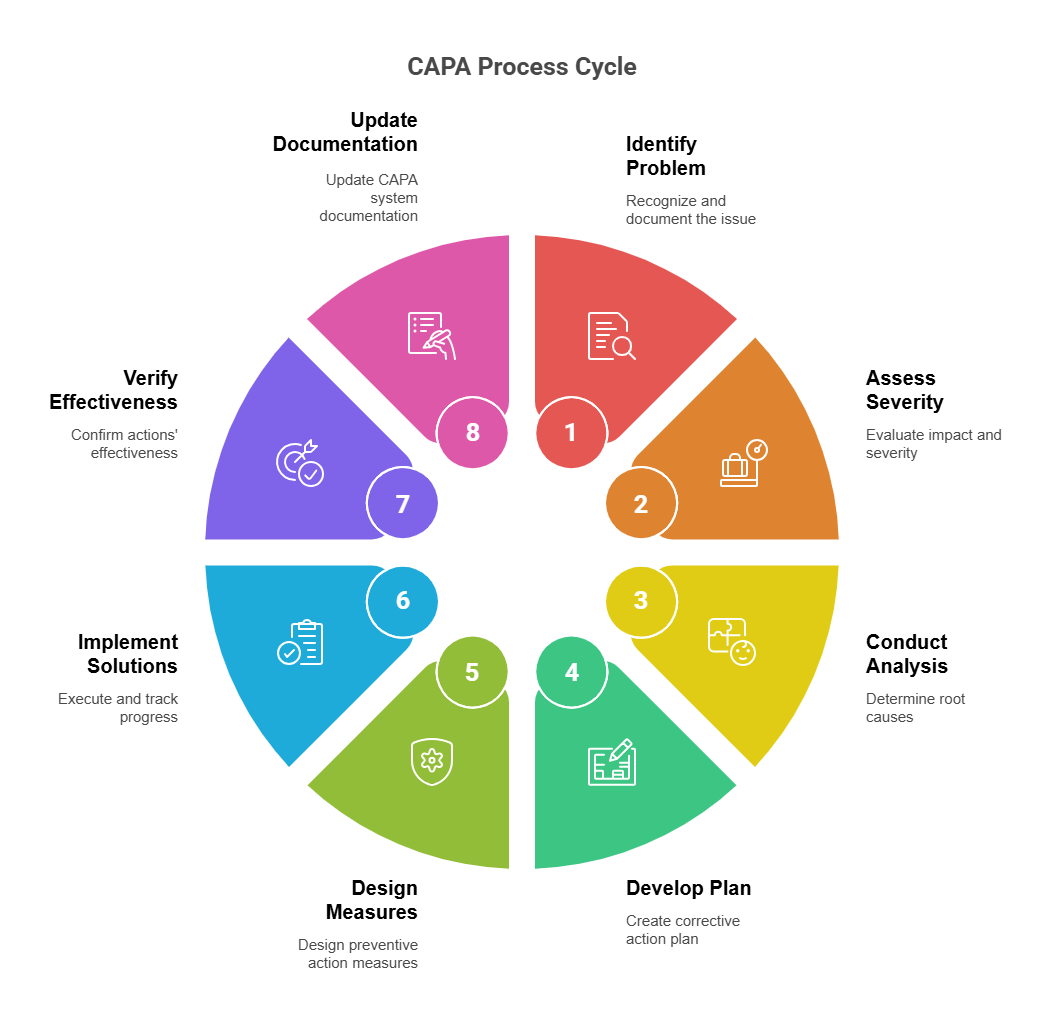
3. CAPA (Corrective and Preventive Action)
4
CAPA ensures continuous improvement.
- Corrective Action: Fix current issue
- Preventive Action: Prevent recurrence
4. Change Control System
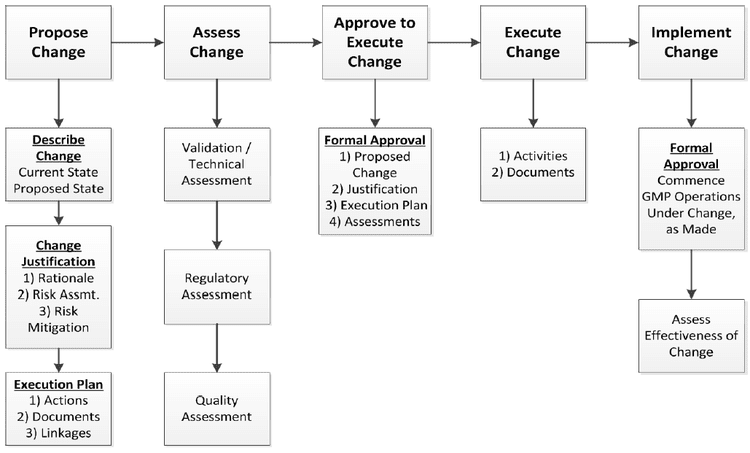
Manages changes in:
- Equipment
- Process
- Raw materials
- SOPs
Ensures no negative impact on product quality.
5. Validation & Qualification

Types:
- Process Validation
- Cleaning Validation
- Equipment Qualification (IQ, OQ, PQ)
6. Training Management

4
Ensures employees are competent.
Includes:
- GMP training
- SOP training
- Safety training
7. Internal Audit (Self-Inspection)

Regular audits help identify gaps before regulatory inspections.
8. Risk Management (ICH Q9)

4
Tools:
- FMEA
- HACCP
- Risk matrix
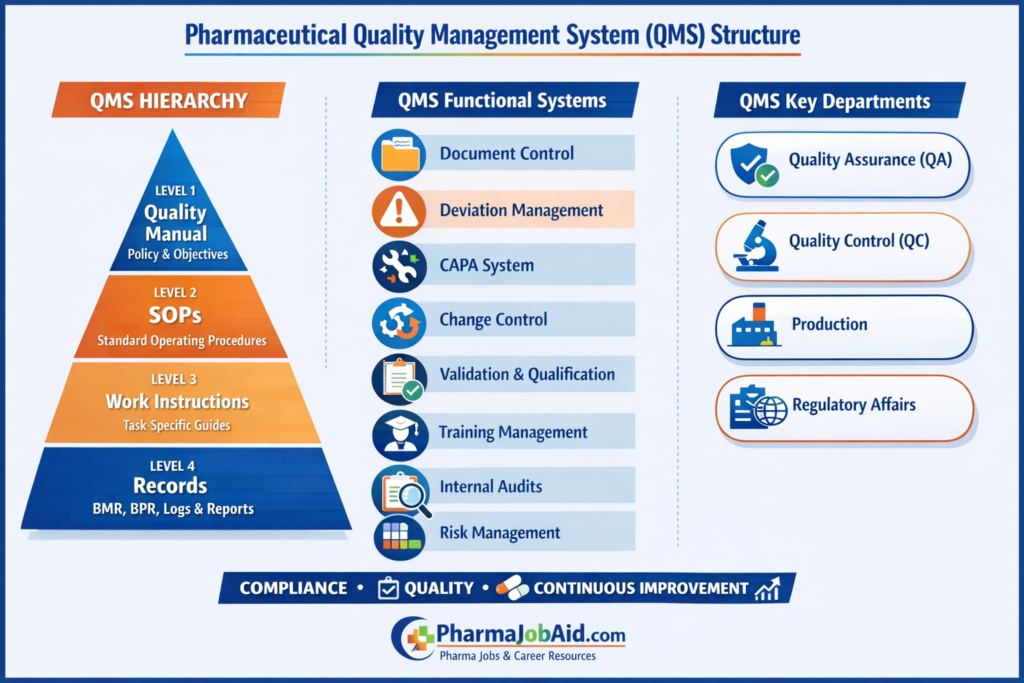
Structure of a Pharmaceutical Quality Management System (QMS)
A Pharmaceutical Quality Management System (QMS) is not just a collection of SOPs—it is a hierarchical, risk-based, and integrated framework aligned with global standards such as International Council for Harmonisation, World Health Organization, and U.S. Food and Drug Administration.
In Bangladesh, this structure is implemented under the supervision of the DGDA and aligned with WHO GMP for both local and export markets.
🧩 Core Structure of a Pharmaceutical QMS

4
The pharmaceutical QMS follows a multi-level hierarchical structure, ensuring control, traceability, and compliance at every stage.
🔷 Level 1: Quality Manual (Strategic Level)
The Quality Manual is the top-level document that defines the company’s overall quality philosophy.
Key Elements:
- Quality Policy
- Quality Objectives
- Organizational Structure
- Scope of QMS
- Regulatory commitments (WHO, FDA, EMA)
Purpose:
- Provides a strategic direction
- Aligns company goals with regulatory expectations
👉 Think of this as the “constitution” of the pharma company’s quality system.
🔷 Level 2: Standard Operating Procedures (SOPs)
SOPs are the backbone of pharmaceutical QMS.
Covers:
- Production processes
- Quality Control testing
- Equipment operation
- Cleaning procedures
- Deviation & CAPA handling
Example SOP Categories:
- SOP for Deviation Management
- SOP for Change Control
- SOP for Line Clearance
- SOP for Validation
Key Features:
- Step-by-step instructions
- Approved by QA
- Controlled documents
🔷 Level 3: Work Instructions & Protocols (Operational Level)
These are detailed, task-specific instructions derived from SOPs.
Includes:
- Equipment operation guidelines
- Analytical test methods
- Validation protocols (IQ/OQ/PQ)
- Calibration procedures
Example:
- HPLC operation instruction
- Tablet compression setup guide
👉 More detailed than SOPs and used directly by operators.
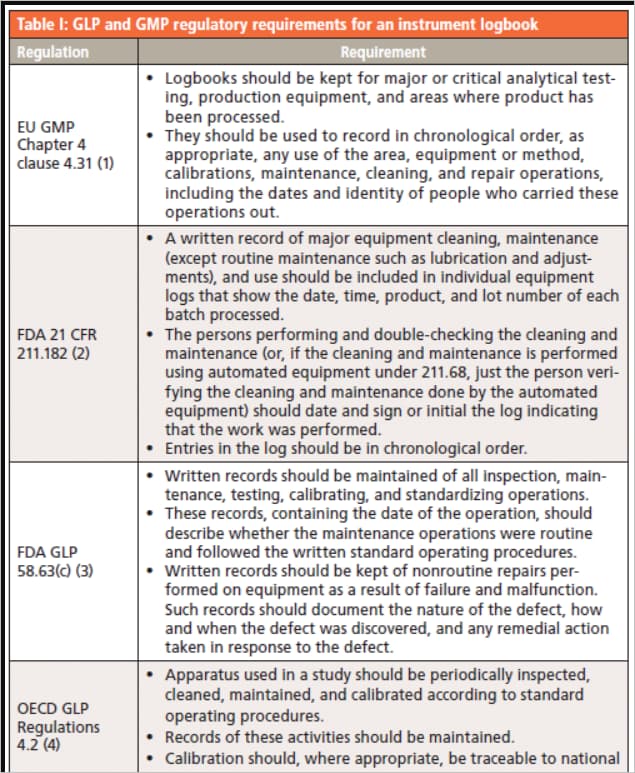
🔷 Level 4: Records & Documentation (Evidence Level)
This is the most critical level for compliance.


4
Includes:
- Batch Manufacturing Records (BMR)
- Batch Packaging Records (BPR)
- Logbooks
- Analytical reports
- Training records
Importance:
- Provides proof of compliance
- Essential during audits and inspections
Must follow:
ALCOA+ principles
- Attributable
- Legible
- Contemporaneous
- Original
- Accurate
🔄 Functional Structure of QMS Systems
Beyond documentation hierarchy, QMS is also structured into functional subsystems:
1. Document Control System
- Creation, review, approval, and distribution of documents
- Version control and archival
2. Deviation Management System
- Identification of deviations
- Root Cause Analysis (RCA)
- Impact assessment
3. CAPA System
- Corrective actions (fix issues)
- Preventive actions (avoid recurrence)
4. Change Control System
- Controls any change in:
- Equipment
- Process
- Materials
- Documents
5. Validation & Qualification System
- Process validation
- Cleaning validation
- Equipment qualification (IQ, OQ, PQ)
6. Training Management System
- Ensures employee competency
- Tracks GMP and SOP training
7. Audit & Self-Inspection System
- Internal audits
- Supplier audits
- Regulatory inspection readiness
8. Quality Risk Management (QRM)
Aligned with ICH Q9:
- Risk identification
- Risk evaluation
- Risk control
Tools:
- FMEA
- Risk matrix
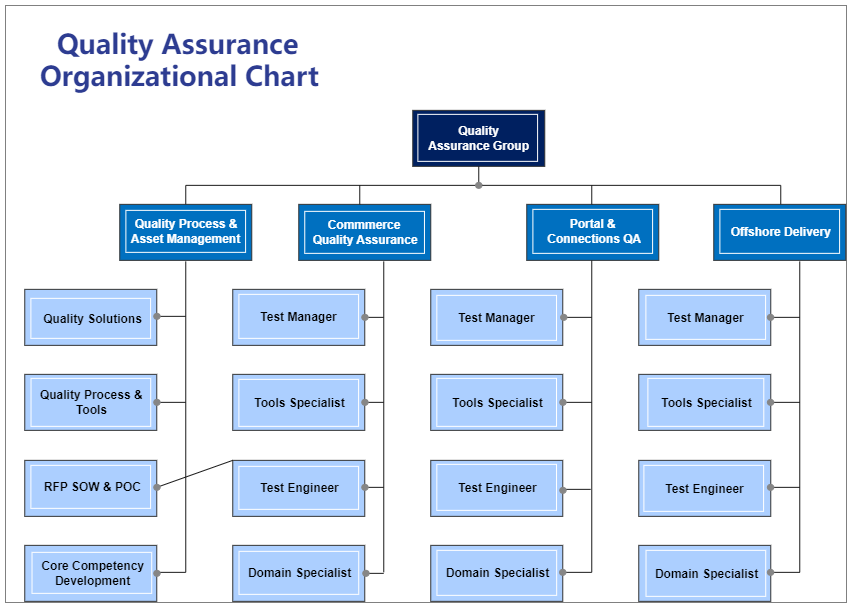
🏗️ Organizational Structure of QMS

4
A pharmaceutical QMS is supported by a cross-functional organizational structure:
Key Departments:
Quality Assurance (QA)
- System owner of QMS
- SOP approval
- Audit & compliance
Quality Control (QC)
- Testing of raw materials, in-process, finished goods
Production
- Manufacturing operations
Engineering
- Equipment maintenance & qualification
Regulatory Affairs
- Compliance with global regulations
👉 QA must remain independent from production (as per GMP).
🔁 QMS Process Flow Structure
A typical pharmaceutical QMS operates in a continuous improvement loop:
- Define Quality Policy
- Develop SOPs
- Execute processes
- Record data
- Monitor performance
- Identify deviations
- Implement CAPA
- Improve system
📊 Integration with ICH Q10 Model
According to International Council for Harmonisation, QMS structure integrates:
- Process Performance & Product Quality Monitoring
- CAPA System
- Change Management System
- Management Review
🇧🇩 Special Considerations in Bangladesh
In Bangladesh, QMS structure is influenced by:
- DGDA regulatory framework
- WHO GMP compliance
- Export market requirements
Industry Leaders Applying Strong QMS:
- Square Pharmaceuticals
- Beximco Pharma
- Incepta Pharmaceuticals
🚀 Best Practices for Structuring QMS
- Maintain clear hierarchy (Manual → SOP → Records)
- Ensure document control discipline
- Implement risk-based approach
- Digitize using eQMS
- Train employees regularly
