Standard Operating Procedures (SOPs) in the Pharmaceutical Industry
Introduction
The pharmaceutical industry in Bangladesh has transformed into one of the most dynamic sectors in South Asia, contributing significantly to the national economy and healthcare system. With local manufacturers supplying nearly 97% of domestic demand and exporting to over 150 countries, maintaining international quality standards is no longer optional—it is mandatory.
At the heart of this quality system lies one critical element: Standard Operating Procedures (SOPs).
SOPs are not just documents—they are the operational backbone that ensures every task, from raw material handling to final product release, is executed consistently, safely, and in compliance with regulatory standards such as GMP (Good Manufacturing Practice).
For pharmaceutical professionals in Bangladesh—whether in Quality Assurance (QA), Quality Control (QC), Production, or Regulatory Affairs—understanding SOPs is essential for both compliance and career growth.
👉 Explore pharma career opportunities: https://pharmajobaid.com/
What is a Standard Operating Procedure (SOP)?
A Standard Operating Procedure (SOP) is a detailed, written instruction designed to achieve uniformity in performing specific functions. In the pharmaceutical industry, SOPs define how tasks must be performed to ensure product quality, safety, and compliance.
Key Features of SOPs:
- Step-by-step instructions
- Clear responsibilities
- Regulatory alignment
- Document control
- Repeatability and consistency
SOPs ensure that even complex pharmaceutical processes can be performed consistently by different personnel without compromising quality.
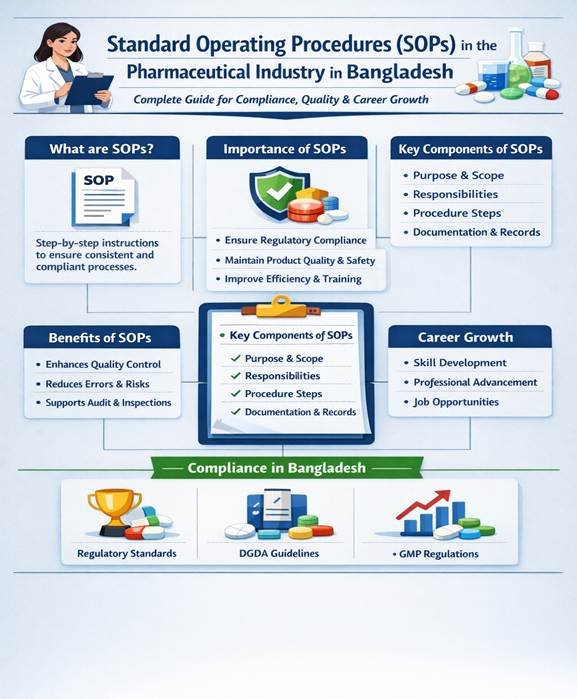
Importance of SOPs in the Pharmaceutical Industry in Bangladesh
In Bangladesh, pharmaceutical companies must comply with strict regulatory standards set by authorities such as the Directorate General of Drug Administration (DGDA) and international bodies like WHO and ICH.
1. Ensuring GMP Compliance
SOPs are essential for implementing GMP guidelines, which require:
- Controlled processes
- Documented procedures
- Traceability
Without SOPs, GMP compliance is impossible.
🔗 WHO GMP Guidelines: https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/gmp
2. Maintaining Product Quality
Consistency is the key to pharmaceutical quality. SOPs ensure that:
- Each batch is produced under identical conditions
- Variability is minimized
- Product safety is maintained
3. Regulatory Inspection Readiness
Regulatory inspections by DGDA or international agencies focus heavily on SOPs. Inspectors check:
- Availability of SOPs
- Proper implementation
- Employee understanding
4. Minimizing Human Error
Human error is one of the biggest risks in pharmaceutical manufacturing. SOPs reduce this risk by:
- Providing clear instructions
- Standardizing processes
- Eliminating guesswork
5. Training and Knowledge Transfer
SOPs act as training tools for new employees and ensure continuity of knowledge across teams.
👉 Read QA career guide: https://pharmajobaid.com/qa-pharma/
Types of SOPs in Pharmaceutical Industry
SOPs vary depending on the department and function.
1. Quality Assurance (QA SOPs)
QA SOPs ensure compliance with regulatory standards and internal quality systems.
Examples:
- Change control SOP
- Deviation management SOP
- CAPA SOP
- Document control SOP
QA acts as the guardian of SOP systems.
2. Quality Control (QC SOPs)
QC SOPs focus on testing and analysis of raw materials, intermediates, and finished products.
Examples:
- Sampling SOP
- Analytical testing SOP
- Stability testing SOP
- Instrument calibration SOP
👉 Explore QC roles: https://pharmajobaid.com/qc-pharma-industry/
3. Production SOPs
Production SOPs define manufacturing processes.
Examples:
- Batch manufacturing procedure
- Equipment operation SOP
- Cleaning SOP
- Line clearance SOP
4. Warehouse SOPs
Warehouse SOPs ensure proper storage and handling.
Examples:
- Raw material receiving
- Storage conditions
- Inventory control
- Dispatch procedures
5. Engineering SOPs
Engineering SOPs maintain equipment and utilities.
Examples:
- Preventive maintenance
- HVAC system operation
- Water system validation
6. Regulatory Affairs SOPs
These SOPs ensure compliance with global regulatory requirements.
Examples:
- Dossier preparation
- Submission process
- Variation handling
Detailed Structure of a Pharmaceutical SOP
A well-structured Standard Operating Procedure (SOP) is critical for ensuring clarity, compliance, and audit readiness in the pharmaceutical industry. Regulatory bodies such as the World Health Organization (WHO), FDA, and DGDA expect SOPs to follow a standardized format that ensures traceability, accountability, and consistency.
In Bangladesh, pharmaceutical companies—especially export-oriented manufacturers—must maintain SOP structures aligned with global GMP expectations. A poorly structured SOP can lead to audit observations, batch rejection, or even regulatory action.
Below is a complete, industry-grade SOP structure used in pharmaceutical companies.
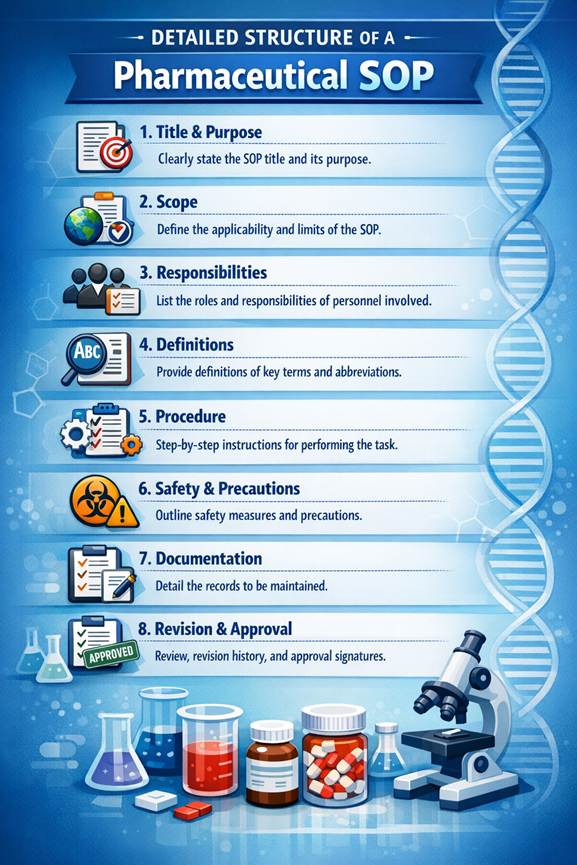
1. SOP Header Section (Document Control Block)
The header is the most critical section for document control and traceability.
Key Elements:
- Company Name & Logo
- Department Name (e.g., Quality Assurance, Production)
- SOP Title (Clear and specific)
- SOP Number / Code (Unique identifier)
- Version Number (e.g., Version 01, Rev. 02)
- Effective Date
- Review Date
- Supersedes (Previous SOP version reference)
- Page Numbering (Page X of Y)
Example:
SOP No: QA-001
Title: Cleaning Procedure for Tablet Compression Machine
Version: 02
Effective Date: 01-Jan-2026
Page: 1 of 12
👉 Why it matters:
This section ensures document control, prevents use of obsolete SOPs, and supports audit traceability.
2. Purpose / Objective
This section explains why the SOP exists.
Best Practices:
- Keep it short and precise
- Clearly state the goal
- Avoid unnecessary details
Example:
“To establish a standardized procedure for cleaning tablet compression machines to prevent cross-contamination and ensure compliance with GMP.”
👉 SEO Tip: Include keywords like SOP in pharmaceutical industry, GMP compliance, cleaning procedure pharma.
3. Scope
The scope defines where and when the SOP applies.
Include:
- Departments covered
- Equipment/process included
- Exclusions (if any)
Example:
“This SOP applies to all tablet compression machines in the production department of XYZ Pharmaceuticals Ltd.”
👉 Why important: Prevents misuse or misinterpretation of SOP.
4. Responsibility
Defines who is responsible for what.
Typical Roles:
- Operator
- Supervisor
- QA Officer
- Engineering Team
Example:
- Operator: Perform cleaning as per SOP
- Supervisor: Verify completion
- QA: Approve and monitor compliance
👉 Audit Insight: Inspectors often check whether responsibilities are clearly defined.
5. Definitions and Abbreviations
Clarifies technical terms used in the SOP.
Examples:
- SOP = Standard Operating Procedure
- GMP = Good Manufacturing Practice
- QA = Quality Assurance
- QC = Quality Control
👉 Why needed: Ensures uniform understanding across departments.
6. Materials and Equipment
Lists all required materials and tools.
Include:
- Cleaning agents
- Equipment/tools
- Safety gear
Example:
- Purified water
- Detergent solution
- Lint-free cloth
- PPE (gloves, mask)
👉 Compliance Tip: Helps ensure no unauthorized materials are used.
7. Safety Precautions
Critical for protecting personnel and products.
Include:
- PPE requirements
- Hazard warnings
- Emergency procedures
Example:
- Wear gloves and mask during cleaning
- Avoid direct contact with chemicals
👉 GMP Requirement: Safety must always be integrated into SOPs.
8. Detailed Procedure (Core Section)
This is the most important part of the SOP.
Best Practices:
- Use step-by-step numbering
- Use clear, simple language
- Avoid ambiguity
- Include critical control points
Example Structure:
- Switch off the machine
- Disconnect power supply
- Remove product residues
- Clean with detergent
- Rinse with purified water
- Dry with lint-free cloth
- Record cleaning in logbook
👉 Advanced Tip:
Highlight critical steps using bold or warnings:
⚠ Ensure no residue remains before starting next batch.
9. In-Process Controls (if applicable)
Defines checks during execution.
Examples:
- Visual inspection
- pH check
- Temperature monitoring
👉 Why important: Ensures process consistency and quality control.
10. Documentation and Records
Specifies what records must be maintained.
Include:
- Logbooks
- Batch records
- Cleaning records
Example:
“All cleaning activities must be recorded in the equipment cleaning logbook.”
👉 Audit Focus: Documentation is a key inspection area.
11. Attachments / Annexures
Includes supporting documents.
Examples:
- Checklists
- Forms
- Log sheets
- Diagrams
👉 Example:
- Annexure I: Cleaning Checklist
- Annexure II: Logbook Format
12. References
Lists regulatory and internal references.
Examples:
- WHO GMP Guidelines
- FDA regulations
- ICH Q7
👉 External References:
- WHO GMP: https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/gmp
- FDA: https://www.fda.gov/drugs/pharmaceutical-quality-resources
13. Revision History
Tracks SOP updates.
Example Table:
| Version | Date | Changes Made |
| 01 | Jan 2025 | Initial issue |
| 02 | Jan 2026 | Updated procedure |
👉 Why important: Ensures version control and audit traceability.
14. Approval Section
Confirms SOP authorization.
Include:
- Prepared by
- Reviewed by
- Approved by
With:
- Name
- Signature
- Date
👉 Compliance Insight: SOP is invalid without proper approval.
15. Distribution List (Optional but Recommended)
Specifies where SOP copies are distributed.
Example:
- QA Department
- Production Floor
- Document Control Room
👉 Prevents unauthorized or outdated copies.
16. Training Requirement Section
Defines training needs before implementation.
Include:
- Training type
- Responsible trainer
- Documentation
👉 Example:
“All operators must be trained on this SOP before execution.”
17. Change Control Reference
Links SOP updates to change control system.
👉 Example:
“Any modification to this SOP must be processed through the change control system.”
Common Mistakes in SOP Structure
Avoid these errors:
- Missing version control
- Unclear responsibilities
- No approval signatures
- Overly complex language
- Lack of documentation section
Pro Tips for Bangladesh Pharma Industry
- Align SOPs with DGDA + WHO GMP
- Use simple English for workforce clarity
- Maintain strict document control system
- Regularly review SOPs (every 1–2 years)
Conclusion
A well-structured SOP is not just a regulatory requirement—it is a quality assurance tool, training manual, and compliance document all in one. In Bangladesh’s rapidly growing pharmaceutical industry, mastering SOP structure is essential for both organizational success and individual career growth.
SOP Lifecycle Management
SOP Lifecycle Management in Pharmaceutical Industry (Bangladesh GMP Perspective)
Standard Operating Procedures (SOPs) are not static documents—they are dynamic, controlled documents that evolve throughout their lifecycle. Effective SOP lifecycle management ensures that procedures remain accurate, compliant, and aligned with current regulatory requirements such as GMP (Good Manufacturing Practice), DGDA guidelines, and international standards like WHO and ICH.
In the pharmaceutical industry in Bangladesh, improper SOP lifecycle management can lead to audit observations, product recalls, batch failures, and regulatory penalties. Therefore, companies must establish a robust SOP lifecycle system governed by Quality Assurance (QA).
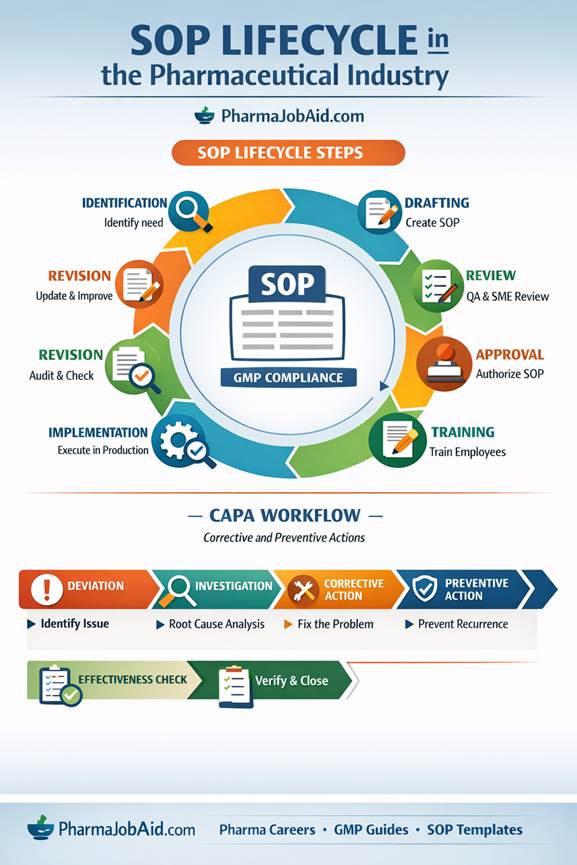
What is SOP Lifecycle Management?
SOP lifecycle management refers to the systematic process of creating, reviewing, approving, implementing, maintaining, and retiring SOPs in a controlled manner.
It ensures:
- Document integrity
- Regulatory compliance
- Continuous improvement
- Version control
- Audit readiness
Key Stages of SOP Lifecycle
A typical SOP lifecycle in pharmaceutical companies consists of the following stages:
1. SOP Identification (Need Assessment)
The lifecycle begins with identifying the need for a new SOP or revision.
When is a new SOP required?
- Introduction of new equipment or process
- Regulatory requirement updates
- Audit observations or CAPA
- Process deviations
- Technology upgrades
Example:
If a new tablet compression machine is installed, a new SOP must be created for its operation and cleaning.
👉 Best Practice:
QA should maintain an SOP master list to track all required SOPs.
2. SOP Drafting (Creation Stage)
Once the need is identified, the SOP is drafted.
Responsible:
- Subject Matter Expert (SME)
- Department Head
Key Considerations:
- Use clear, simple language
- Follow standard SOP format
- Include step-by-step procedures
- Align with GMP requirements
Common Mistakes:
- Overcomplicated language
- Missing steps
- Lack of clarity
👉 Pro Tip:
Use real operational steps—not theoretical descriptions.
3. SOP Review
The draft SOP undergoes a detailed review.
Reviewers:
- Quality Assurance (QA)
- Cross-functional teams (Production, QC, Engineering)
Review Focus:
- Accuracy
- GMP compliance
- Clarity and usability
- Risk assessment
QA Role:
QA ensures the SOP complies with:
- WHO GMP
- DGDA requirements
- Internal quality systems
4. SOP Approval
After review, the SOP is formally approved.
Approval Authorities:
- Department Head
- QA Head
- Quality Director / Authorized Person
Requirements:
- Signature
- Date
- Version control
👉 Important:
An SOP is not valid unless approved.
5. SOP Issuance and Distribution
Once approved, the SOP is issued and distributed.
Distribution Methods:
- Controlled hard copies
- Electronic Document Management System (EDMS)
Key Control Measures:
- Only authorized copies are distributed
- Obsolete versions are removed
- Master copy is maintained by QA
👉 Audit Tip:
Inspectors often check whether obsolete SOPs are still in use.
6. SOP Training
Before implementation, all relevant personnel must be trained.
Training Types:
- Classroom training
- On-the-job training
- E-learning
Documentation:
- Training attendance sheet
- Assessment results
- Training records
Requirement:
No employee should perform a task without SOP training.
👉 Compliance Insight:
Training records are one of the most frequently audited documents.
7. SOP Implementation
After training, the SOP is implemented in operations.
Key Points:
- Follow SOP strictly
- Record all activities
- Monitor compliance
QA Role:
- Ensure SOP adherence
- Conduct periodic checks
8. SOP Monitoring and Compliance
Continuous monitoring ensures SOP effectiveness.
Monitoring Methods:
- Internal audits
- Line inspections
- Process verification
Indicators:
- Deviation frequency
- Error rates
- Compliance level
9. SOP Deviation Handling
If SOP is not followed, it results in a deviation.
Types of Deviations:
- Minor
- Major
- Critical
Process:
- Identify deviation
- Investigate root cause
- Implement corrective action
- Document findings
10. CAPA (Corrective and Preventive Action)
CAPA ensures that issues do not recur.
Steps:
- Root cause analysis
- Corrective action
- Preventive action
- Effectiveness check
👉 Example:
Deviation: Improper cleaning
CAPA: Retrain operator + revise SOP
11. SOP Periodic Review
SOPs must be reviewed regularly.
Frequency:
- Every 1–2 years
- Or as required
Review Triggers:
- Regulatory updates
- Process changes
- Audit findings
12. SOP Revision / Change Control
Any change in SOP must follow a change control system.
Steps:
- Change request
- Impact assessment
- Approval
- SOP update
- Training
👉 Important:
Unauthorized changes are strictly prohibited.
13. SOP Archiving and Retention
Old SOPs must be archived.
Requirements:
- Maintain historical records
- Ensure traceability
- Store securely
Retention Period:
- As per regulatory requirement (often 5–10 years)
14. SOP Obsolescence (Retirement)
When an SOP is no longer required:
- Mark as obsolete
- Remove from circulation
- Archive properly
SOP Lifecycle Flow (Simple Summary)
- Identification
- Drafting
- Review
- Approval
- Distribution
- Training
- Implementation
- Monitoring
- Deviation & CAPA
- Review
- Revision
- Archiving
Importance of SOP Lifecycle Management in Bangladesh Pharma
Effective lifecycle management ensures:
- GMP compliance
- Audit readiness
- Product quality
- Regulatory approval
- Export eligibility
Common SOP Lifecycle Mistakes
- Using outdated SOPs
- Lack of training records
- Poor version control
- Missing approval signatures
- No periodic review
Digital SOP Lifecycle (Modern Trend)
Many pharma companies in Bangladesh are adopting digital systems.
Benefits:
- Real-time updates
- Electronic signatures
- Audit trails
- Reduced paperwork
Role of QA in SOP Lifecycle
Quality Assurance is the owner of SOP lifecycle.
Responsibilities:
- SOP control
- Training oversight
- Audit readiness
SOP Writing Best Practices
Writing an effective SOP requires skill and clarity.
Key Guidelines:
- Use simple language
- Avoid ambiguity
- Use numbered steps
- Include safety precautions
- Maintain consistency
Regulatory Requirements for SOPs in Bangladesh
DGDA Requirements
DGDA mandates:
- Documented procedures
- Record keeping
- Traceability
WHO GMP Requirements
WHO requires:
- Written procedures
- Controlled documents
- Regular updates
ICH Guidelines
ICH ensures global standardization for export markets.
🔗 FDA Reference: https://www.fda.gov/drugs/pharmaceutical-quality-resources
SOPs and GMP Compliance
SOPs are essential for implementing GMP systems.
GMP Areas Covered:
- Documentation
- Personnel hygiene
- Equipment validation
- Process control
Without SOPs, GMP cannot function effectively.
Common SOP Examples in Pharma Industry
Equipment Cleaning SOP
Prevents cross-contamination.
Line Clearance SOP
Ensures no mix-up between batches.
Deviation SOP
Handles unexpected events.
CAPA SOP
Prevents recurrence of issues.
Deviation and CAPA in SOP System
What is Deviation?
A deviation is any departure from an approved SOP.
Types:
- Minor deviation
- Major deviation
- Critical deviation
What is CAPA?
CAPA stands for Corrective and Preventive Action.
- Corrective action: Fix the issue
- Preventive action: Prevent recurrence
SOP Training System
Training is mandatory in pharmaceutical companies.
Types of Training:
- Induction training
- On-the-job training
- Refresher training
Documentation:
- Training records
- Assessment results
SOP Audit and Inspection
Audits ensure compliance.
Audit Focus Areas:
- SOP availability
- Implementation
- Employee knowledge
Challenges in SOP Implementation in Bangladesh
1. Lack of Awareness
Some employees do not fully understand SOP importance.
2. Poor Documentation
Incomplete records create compliance issues.
3. Resistance to Change
Employees resist updated SOPs.
4. Resource Constraints
Smaller companies face limitations.
Digital SOP Systems in Pharma
Modern companies are shifting to digital SOP systems.
Benefits:
- Electronic documentation
- Real-time updates
- Audit trails
- Better compliance
Role of SOPs in Pharmaceutical Careers
SOP knowledge is essential for:
- QA Officer
- QC Analyst
- Production Executive
- Regulatory Affairs Specialist
👉 Find pharma jobs: https://pharmajobaid.com/jobs-in-pharma-companies/
SOP Interview Questions
- What is SOP?
- Why SOP is important?
- What is deviation?
- Explain CAPA
Case Study: SOP Failure in Pharma Industry
A Bangladeshi pharmaceutical company once faced regulatory action due to improper documentation and failure to follow SOPs in cleaning procedures.
Result:
- Batch rejection
- Financial loss
- Reputation damage
Lesson:
Strict SOP adherence is critical.
Future of SOPs in Bangladesh Pharmaceutical Industry
- Digital transformation
- AI integration
- Global regulatory alignment
- Automation
