Introduction
The pharmaceutical industry continues to evolve at an unprecedented pace, driven by innovation, regulatory advancements, and global healthcare demands. One of the most important indicators of this progress is the number and type of drug approvals granted each year by the U.S. Food and Drug Administration (FDA).
The infographic above highlights new FDA drug approvals through 2025, showcasing both small molecules and biologics across diverse therapeutic areas such as oncology, neurology, cardiology, and infectious diseases.
For professionals, students, and job seekers in the pharmaceutical industry, understanding these approvals is more than academic—it directly impacts career opportunities, research directions, and industry growth trends.
👉 Explore more career insights and pharma job opportunities at
https://pharmajobaid.com/
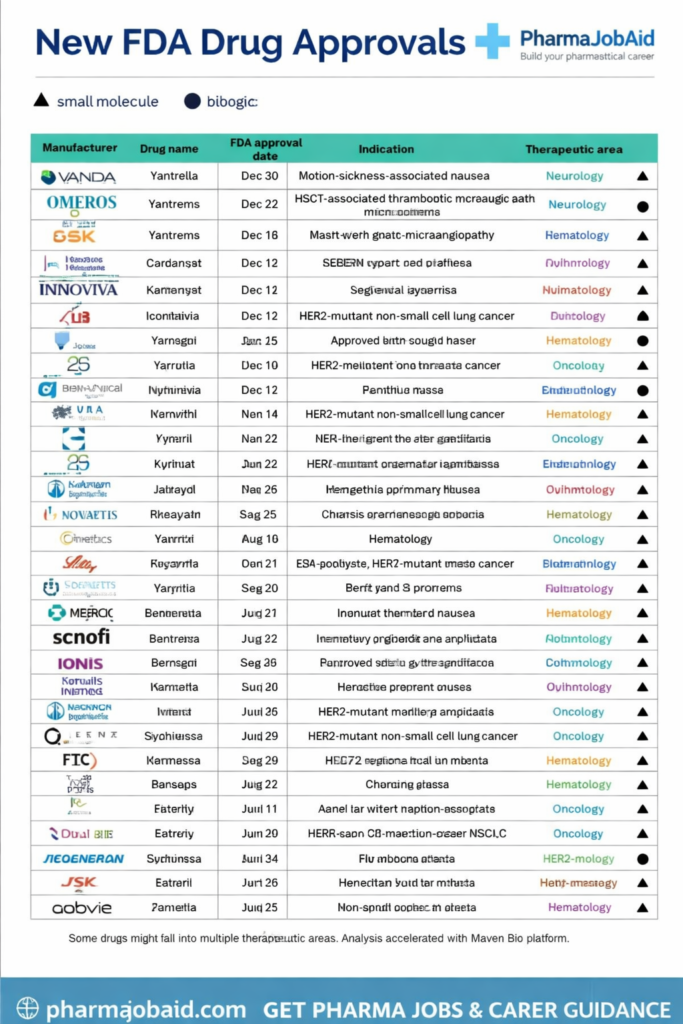
Understanding FDA Drug Approvals
What is FDA Drug Approval?
FDA drug approval is the process through which new medications are evaluated for safety, efficacy, and quality before being made available to the public.
The approval process includes:
- Preclinical studies
- Clinical trials (Phase I, II, III)
- New Drug Application (NDA) or Biologics License Application (BLA)
- Regulatory review and inspection
- Post-marketing surveillance
Why FDA Approvals Matter
FDA approvals are critical because they:
- Ensure patient safety
- Introduce innovative treatments
- Drive pharmaceutical market growth
- Create new job roles in pharma companies
Overview of FDA Drug Approvals Through 2025
The infographic reveals a diverse portfolio of newly approved drugs categorized into:
1. Small Molecules
- Chemically synthesized drugs
- Typically oral dosage forms
- Cost-effective and widely used
2. Biologics
- Derived from living organisms
- Include monoclonal antibodies, gene therapies
- Highly targeted and complex
Key Trends in FDA Drug Approvals
1. Rise of Oncology Drugs
A major portion of approvals focuses on cancer treatment, including:
- HER2-mutant cancers
- Breast cancer
- Multiple myeloma
- Non-small cell lung cancer (NSCLC)
Why Oncology Dominates
- High global disease burden
- Rapid advancements in targeted therapy
- Increased investment in biotech research
2. Growth of Rare Disease Treatments
Several approvals target rare genetic conditions, including:
- Hereditary angioedema
- Genetic enzyme deficiencies
- Rare metabolic disorders
Industry Insight
Rare disease drugs benefit from:
- Orphan drug incentives
- Faster regulatory pathways
- Premium pricing strategies
3. Expansion of Biologics
Biologics continue to grow rapidly due to:
- Precision medicine
- Immunotherapy breakthroughs
- Advanced biologic manufacturing technologies
4. Focus on Neurology and CNS Disorders
New drugs target:
- Motion sickness
- Neurofibromatosis
- Myasthenia gravis
- Pain management
This indicates increasing investment in central nervous system (CNS) therapies.
5. Increased Infectious Disease Innovation
Post-pandemic awareness has led to:
- RSV prophylaxis drugs
- New antiviral therapies
- Improved vaccination technologies
Therapeutic Areas Breakdown
Oncology
The largest category in the infographic includes drugs for:
- Breast cancer
- Lung cancer
- Leukemia
- Solid tumors
Key Insight
Oncology remains the fastest-growing pharmaceutical segment globally.
Cardiology
New approvals address:
- Hypertrophic cardiomyopathy
- Tachycardia
- Cholesterol management
Industry Trend
Cardiovascular diseases remain a leading cause of mortality, driving continuous innovation.
Neurology
Drugs for:
- Motion sickness
- Neurodegenerative conditions
- Chronic pain
Immunology
Includes treatments for:
- Chronic eczema
- Angioedema
- Autoimmune disorders
Endocrinology
Approvals include therapies for:
- Diabetes-related complications
- Hormonal disorders
- Rare endocrine diseases
Major Pharmaceutical Companies Involved
The infographic features leading global pharmaceutical companies such as:
- Pfizer
- Novartis
- Johnson & Johnson
- Sanofi
- Merck
- AstraZeneca
- GSK
These companies continue to dominate FDA approvals due to:
- Strong R&D pipelines
- Advanced clinical trial capabilities
- Global regulatory expertise
Impact on the Global Pharmaceutical Industry
Market Growth
FDA approvals directly influence:
- Global drug sales
- Investment trends
- Stock market performance
Innovation Acceleration
New approvals encourage:
- Research collaborations
- AI-driven drug discovery
- Personalized medicine
Career Opportunities Created by FDA Approvals
Each new drug approval generates demand for skilled professionals in:
1. Research & Development (R&D)
- Drug discovery scientists
- Clinical researchers
- Biostatisticians
2. Regulatory Affairs
- Regulatory specialists
- Submission experts
- Compliance officers
3. Quality Assurance (QA)
- GMP compliance
- Documentation review
- Audit preparation
4. Quality Control (QC)
- Analytical testing
- Lab validation
- Stability studies
5. Manufacturing
- Production pharmacists
- Process engineers
- Validation experts
6. Pharmacovigilance
- Drug safety monitoring
- Adverse event reporting
- Risk management
👉 Find pharma career opportunities here:
https://pharmajobaid.com/jobs
Skills Required in the Modern Pharma Industry
To succeed in today’s pharmaceutical landscape, professionals need:
Technical Skills
- GMP knowledge
- Regulatory guidelines (FDA, EMA, ICH)
- Analytical techniques
Digital Skills
- Data analytics
- AI in pharma
- Electronic documentation systems
Soft Skills
- Communication
- Problem-solving
- Critical thinking
Role of Regulatory Agencies Beyond FDA
While FDA is the most influential regulator, other agencies include:
- EMA (Europe)
- MHRA (UK)
- PMDA (Japan)
- DGDA (Bangladesh)
These agencies align with global standards to ensure drug safety.
Challenges in Drug Approval
Despite advancements, challenges remain:
1. High Development Costs
- Average cost exceeds $2 billion per drug
2. Long Timelines
- 10–15 years from discovery to approval
3. Regulatory Complexity
- Strict compliance requirements
4. Clinical Trial Failures
- High attrition rates
Future of Drug Approvals
1. AI-Driven Drug Discovery
Artificial intelligence is revolutionizing:
- Target identification
- Molecule design
- Clinical trial optimization
2. Personalized Medicine
Treatments tailored to:
- Genetic profiles
- Biomarkers
- Individual health data
3. Gene and Cell Therapy
Next-generation therapies include:
- CRISPR-based treatments
- CAR-T cell therapy
How PharmaJobAid Helps You Stay Ahead
PharmaJobAid.com is your gateway to a successful pharmaceutical career.
Services Offered
- Job listings in pharma companies
- CV & resume preparation
- Interview guidance
- Career counseling
👉 Visit: https://pharmajobaid.com/
Internal Linking Strategy (For SEO)
You can internally link this article to:
- QA in Pharma
- QC in Pharma Industry
- Pharmaceutical Jobs Guide
- Pharma 4.0 Digital Transformation
External Linking Strategy
Add outbound links to authoritative sources:
- FDA official website (https://www.fda.gov/)
- EMA guidelines
- WHO pharmaceutical standards
- ICH guidelines
Conclusion
The FDA drug approvals through 2025 highlight a rapidly evolving pharmaceutical landscape marked by:
- Strong growth in oncology and biologics
- Increased focus on rare diseases
- Technological advancements in drug development
For professionals, these trends translate into expanding career opportunities and the need for continuous skill development.
Whether you are a student, fresh graduate, or experienced professional, staying updated with FDA approvals can give you a competitive edge in the pharma job market.
Final Call to Action
🚀 Ready to build your pharmaceutical career?
👉 Visit https://pharmajobaid.com/
✅ Explore jobs
✅ Upload your resume
✅ Get career guidance
