Change Control in Pharma: Comprehensive Guide for Compliance and Quality
Table of Contents
- Introduction to Change Control in Pharma
- Types of Change Control in the Pharmaceutical Industry
- Equipment Change Control
- Process Change Control
- Document Change Control
- Regulatory Significance of Change Control
- Out of Trend (OOT) vs Out of Specification (OOS)
- Implementing Effective Equipment Change Control
- Line Clearance in Pharmaceutical Manufacturing
- Integrating OOT/OOS Monitoring with Change Control
- Best Practices for Change Control in Pharma
- Real-World Case Studies
- Advanced Topics in Change Control
- Conclusion
- References
1. Introduction to Change Control in Pharma
In the pharmaceutical industry, patient safety, product quality, and regulatory compliance are non-negotiable. Change control pharma is a structured system that ensures every change in manufacturing processes, equipment, materials, or documentation is assessed, approved, and documented.
A strong change control process:
- Minimizes risk of defective batches
- Ensures compliance with GMP Guidelines
- Maintains consistency in pharmaceutical manufacturing processes
- Provides traceability for audits and inspections
Example: Upgrading a tablet press without proper change control could lead to inconsistent tablet hardness, OOS assay results, and ultimately, regulatory action.
Change control is a core component of the Pharmaceutical Quality System recommended by ICH Q10, and is critical to managing both routine and high-risk changes.
2. Types of Change Control in the Pharmaceutical Industry
Pharmaceutical change control is classified into three primary types: equipment, process, and document. Each type ensures modifications do not compromise product quality or regulatory compliance.
2a. Equipment Change Control
Equipment modifications can include upgrades, replacements, relocation, or calibration changes. Equipment change control ensures these modifications are evaluated for impact on Critical Quality Attributes (CQAs) and Critical Process Parameters (CPPs).
Steps in Equipment Change Control:
- Initiation – Document the proposed change with a clear justification.
- Impact Assessment – Determine potential effects on product quality and GMP compliance.
- Approval – Multi-department approval (QA, Production, Engineering, Validation).
- Validation/Requalification – Conduct IQ, OQ, PQ tests.
- Implementation – Execute the change under controlled conditions.
- Monitoring – Track post-change trends for OOT or OOS deviations.
- Documentation – Maintain full traceability in electronic or physical records.
SOP Example – Equipment Change Control:
SOP ID: EQ-CC-001
Title: Equipment Change Control Procedure
Purpose: Ensure all equipment changes are approved and validated to maintain product quality.
Scope: Applies to all production and QC equipment.
Procedure:
1. Initiate change request with detailed description.
2. Conduct risk assessment evaluating impact on CQAs and CPPs.
3. Obtain approvals from QA, Production, and Engineering.
4. Perform IQ/OQ/PQ validation.
5. Implement change under controlled conditions.
6. Monitor post-change performance; document OOT/OOS deviations.
7. Archive records in the electronic document management system.
Case Example:
A tablet press was upgraded to increase output. OOT trends in tablet hardness were detected post-upgrade. Sensor recalibration and revalidation prevented OOS batch production.
2b. Process Change Control
Process changes involve modifications to manufacturing steps, formulations, or analytical procedures. Examples include:
- Adjusting coating solvents
- Modifying environmental conditions
- Changing raw material suppliers
All changes are assessed for risk and validated to maintain compliance and product quality.
Best Practices:
- Risk assessments using FMEA
- Pilot batch validation
- SOP updates and personnel training
2c. Document Change Control
Document changes include updates to SOPs, batch records, validation protocols, and testing procedures. Proper document change control ensures consistency across all departments and readiness for regulatory inspections.
SOP Example – Document Change Control:
SOP ID: DOC-CC-001
Title: Document Change Control Procedure
Purpose: Ensure all controlled documents reflect approved changes.
Procedure:
1. Identify impacted documents.
2. Revise content with tracked changes.
3. Cross-departmental review and approval.
4. Communicate updates to personnel.
5. Archive previous versions for traceability.
3. Regulatory Significance of Change Control
Regulatory authorities treat poorly managed change control as a critical compliance risk. Adherence to regulations ensures operational integrity, minimizes inspection findings, and protects patient safety.
Key Guidelines:
- FDA 21 CFR Part 211 – Focuses on process and equipment control
- ICH Q10 – Promotes a robust Pharmaceutical Quality System
- EU GMP Guidelines – Annex 15 – Emphasizes qualification and validation
Audit Focus Areas:
- Documentation of all changes
- Risk assessment reports
- Validation outcomes after changes
- OOT/OOS investigation records
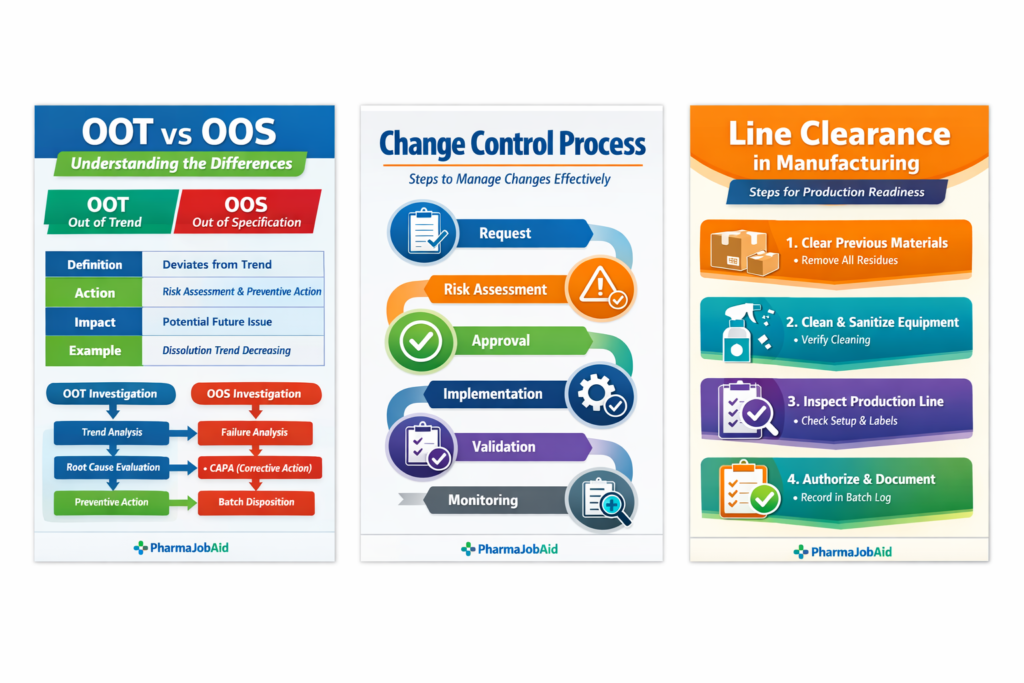
4. Out of Trend (OOT) vs Out of Specification (OOS)
Understanding OOT vs OOS is critical for proactive quality control.
Out of Specification (OOS)
Occurs when test results fall outside acceptance criteria.
- Example: Tablet assay < 95% label claim
- Investigation: Immediate batch quarantine, root cause analysis, CAPA
Out of Trend (OOT)
Results within specification but showing deviation from historical trends.
- Example: Six batches showing decreasing dissolution values
- Investigation: Risk assessment and preventive action
OOT vs OOS Table:
| Feature | OOS | OOT |
|---|---|---|
| Definition | Fails acceptance criteria | Within spec but deviates from trend |
| Action | Batch rejection and investigation | Risk assessment and preventive action |
| Impact | Immediate | Potential future impact |
| Example | API content < 95% | Dissolution trend decreasing |
Infographic Placeholder:
- Visual chart comparing OOT vs OOS investigation workflows
5. Implementing Effective Equipment Change Control
A strong equipment change control program reduces risk of OOT/OOS results:
- Risk assessment on CQAs and CPPs
- Validation/Requalification (IQ/OQ/PQ)
- Personnel training
- Post-change monitoring and OOT/OOS evaluation
- Complete documentation
Workflow Infographic Placeholder:
- Request → Risk Assessment → Approval → Implementation → Validation → Monitoring → Documentation
6. Line Clearance in Pharmaceutical Manufacturing
Line clearance manufacturing prevents cross-contamination and ensures correct product flow.
Steps:
- Remove previous batch materials
- Clean and sanitize equipment
- Inspect line readiness
- Authorization and documentation
SOP Example – Line Clearance:
SOP ID: LC-001
Title: Line Clearance Procedure
Purpose: Prevent cross-contamination between batches.
Procedure:
1. Verify previous batch removal.
2. Clean and sanitize equipment.
3. Inspect and approve line readiness.
4. Document completion in batch record.
Infographic Placeholder:
- Stepwise diagram of line clearance in manufacturing
7. Integrating OOT/OOS Monitoring with Change Control
OOT/OOS data informs change control decisions:
- Early detection of trends prevents OOS batches
- Supports risk-based decisions on equipment maintenance
- Drives continuous process improvement
Best Practices:
- Implement electronic batch records
- Use statistical process control (SPC)
- Document and review all investigations
8. Best Practices for Change Control in Pharma
- Document all changes comprehensively
- Conduct risk assessments using FMEA or HACCP
- Train personnel on procedures and OOT/OOS significance
- Leverage digital systems for tracking changes
- Conduct periodic audits for compliance and efficiency
9. Real-World Case Studies
Case Study 1: Tablet Press Upgrade
- Equipment change led to OOT trends
- Corrective action: sensor recalibration
- Outcome: Compliance maintained
Case Study 2: Coating Solvent Change
- Process change caused OOS dissolution
- CAPA implemented, SOP updated
- Regulatory notification completed
Case Study 3: Line Clearance Failure
- Improper line clearance detected during audit
- CAPA: retraining, revised SOP, verification checklist
- Outcome: Improved compliance and zero contamination
10. Advanced Topics in Change Control
- Digital Transformation: Automated change control workflows reduce human error
- Risk-Based Approach: Apply intensive review for high-impact changes
- Predictive Analytics: AI detects early OOT trends and prevents OOS outcomes
11. Conclusion
Effective change control pharma systems:
- Safeguard product quality and patient safety
- Ensure compliance with global standards
- Reduce the risk of OOS batches and OOT deviations
Integration of equipment, process, and document change control with OOT/OOS monitoring ensures robust manufacturing operations.
