📌 Introduction
In today’s highly regulated pharmaceutical environment, maintaining product quality and regulatory compliance is not optional—it is mandatory. One of the most powerful tools supporting this objective is Trend Analysis within the Quality Management System (QMS).
Trend analysis transforms raw data into meaningful insights, helping pharmaceutical companies identify risks, prevent failures, and ensure continuous improvement. Regulatory bodies such as the World Health Organization, U.S. Food and Drug Administration, and European Medicines Agency strongly emphasize data trending as part of GMP compliance.
This guide is designed for professionals, fresh graduates, and industry experts working in Bangladesh’s growing pharmaceutical sector.
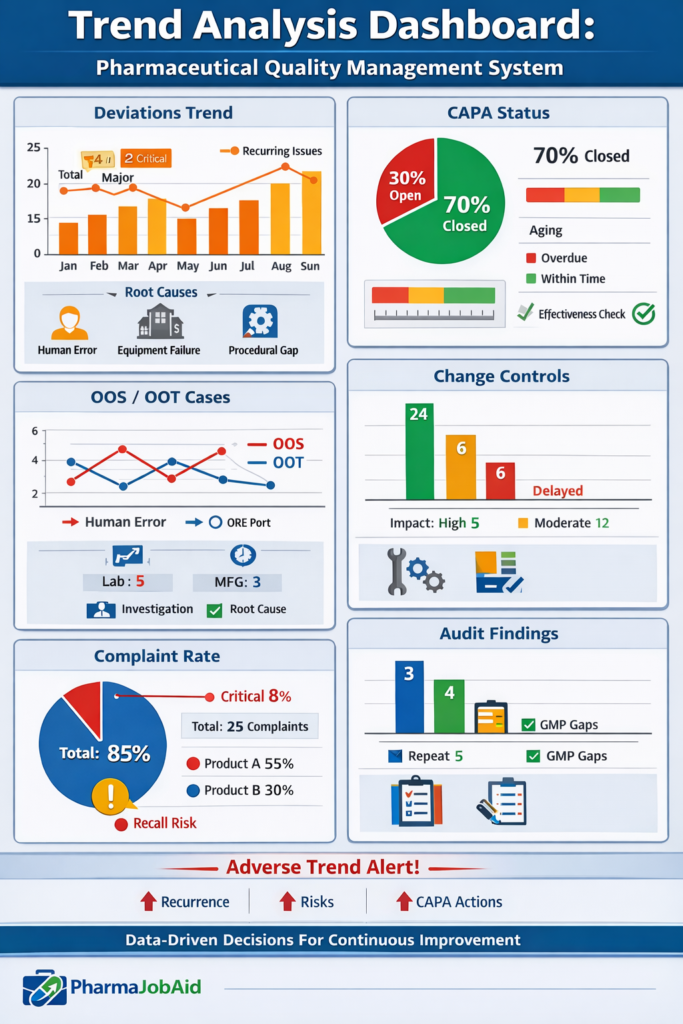
🔍 What is Trend Analysis in Pharmaceutical QMS?
Trend analysis is a systematic process of evaluating quality data over time to:
- Identify recurring issues
- Detect process variability
- Monitor system performance
- Predict potential risks
It applies across all QMS elements, including:
- Deviations
- CAPA
- OOS / OOT
- Change controls
- Complaints
- Audit findings
🏭 Importance of Trend Analysis in Pharma Industry

4
1. Ensures GMP Compliance
Trend analysis is required under global GMP frameworks such as:
- WHO TRS 961
- EU GMP Part I
- ICH Q10
2. Early Detection of Risks
By identifying patterns early, companies can:
- Prevent batch failures
- Reduce recalls
- Improve patient safety
3. Supports Continuous Improvement
Trend data helps refine:
- Processes
- SOPs
- Training systems
4. Data-Driven Decision Making
Management decisions become:
- Objective
- Evidence-based
- Risk-focused
📊 Key QMS Elements for Trending
1. Deviation Trending
- Frequency of deviations
- Root cause analysis
- Recurrence patterns
2. CAPA Trending
- Number of CAPAs raised vs closed
- Effectiveness checks
- Aging trends
3. OOS / OOT Analysis
- Laboratory vs manufacturing errors
- Analytical method variability
- Stability failures
4. Change Control Trends
- Number of changes
- Impact levels
- Delayed closures
5. Complaint Trending
- Market complaints by product
- Severity classification
- Root cause trends
6. Audit Findings
- Internal vs external audits
- Repeat observations
- Compliance gaps
📈 Types of Trend Analysis Used in Pharma


4
✔️ Quantitative Analysis
- Number of events per month
- Rate per batch
- Department-wise distribution
✔️ Qualitative Analysis
- Root cause evaluation
- Risk classification
- Process impact
📉 Statistical Tools Used in Trend Analysis
Pharmaceutical companies commonly use:
- Line Charts → Track performance over time
- Bar Charts → Compare categories
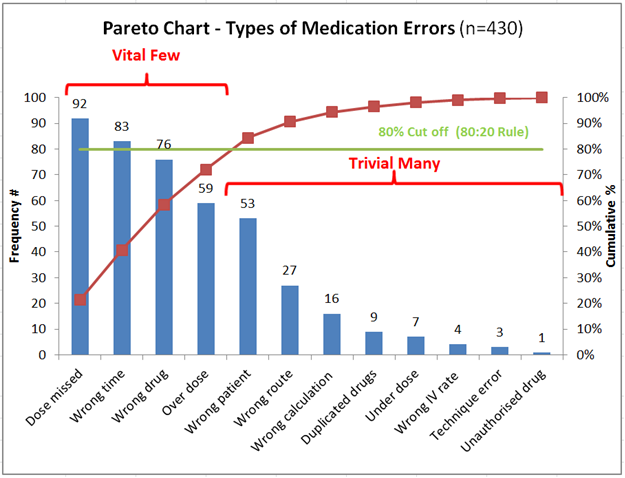
- Pareto Charts → Identify major contributors (80/20 rule)
- Control Charts → Monitor process stability
- Moving Averages → Smooth data variations
🔄 Trend Analysis Workflow (Step-by-Step)
Step 1: Data Collection
Gather data from:
- Deviation logs
- CAPA tracker
- OOS reports
- Audit reports
Step 2: Data Classification
Categorize based on:
- Type
- Department
- Severity
Step 3: Data Analysis
Use statistical tools to:
- Identify patterns
- Detect abnormal spikes
Step 4: Interpretation
Determine:
- Root causes
- Risk levels
Step 5: Action (CAPA)
Implement:
- Corrective actions
- Preventive measures
Step 6: Review
- Management review meetings (MRM)
- Continuous monitoring
🚨 Identifying Adverse Trends
A trend is considered adverse when:
- Frequency increases over time
- Same root cause repeats
- Product quality is impacted
- Regulatory risk is high
🇧🇩 Trend Analysis in Bangladesh Pharmaceutical Industry



4
Bangladesh’s pharmaceutical sector is rapidly expanding with companies like:
- Beximco Pharmaceuticals
- Square Pharmaceuticals
- Incepta Pharmaceuticals
Trend analysis plays a key role in:
- Export compliance (US FDA, EU GMP)
- Maintaining product quality
- Competing in global markets
⚠️ Common Challenges in Trend Analysis
- Poor data quality
- Manual data handling errors
- Lack of statistical expertise
- Delayed CAPA implementation
- Incomplete investigations
✅ Best Practices for Effective Trend Analysis
- Use validated software systems
- Ensure real-time data tracking
- Perform monthly trending reviews
- Integrate with risk management (ICH Q9)
- Train staff on data interpretation
🔗 External Regulatory References
- World Health Organization GMP Guidelines
- U.S. Food and Drug Administration 21 CFR 210/211
- International Council for Harmonisation Pharmaceutical Quality System
🎯 Conclusion
Trend analysis is not just a regulatory requirement—it is a strategic tool that drives quality, efficiency, and compliance in the pharmaceutical industry.
For Bangladesh’s growing pharma sector, implementing a robust trend analysis system can:
- Improve global competitiveness
- Ensure regulatory success
- Enhance patient safety

I’ve been looking for a fast and objective news source covering the latest events in Egypt and the Middle East, especially with everything going on right now. I found this website and I really liked their coverage and live updates.
Thought I’d share it here for those interested:
Check it out here
Let me know what you think about it!