Risk Management in the Pharmaceutical Industry in Bangladesh: A Complete Guide for Compliance, Quality & Career Growth
📌 Introduction
Risk management in the pharmaceutical industry is no longer optional—it is a regulatory expectation, a business necessity, and a core component of patient safety. In Bangladesh, where the pharmaceutical sector supplies over 97% of domestic demand and exports to 150+ countries, managing risk effectively is critical for maintaining global standards.
With increasing regulatory scrutiny from authorities like the Directorate General of Drug Administration and international bodies such as World Health Organization, pharmaceutical companies must adopt structured, science-based risk management systems aligned with International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use guidelines—especially ICH Q9 (Quality Risk Management).
This PharmaJobAid guide provides a comprehensive, SEO-optimized overview of risk management in Bangladesh’s pharmaceutical industry, covering frameworks, tools, real-world applications, and career opportunities.
🔍 What is Risk Management in Pharmaceuticals?
Risk management in pharma refers to the systematic process of identifying, assessing, controlling, communicating, and reviewing risks that may impact product quality, patient safety, regulatory compliance, or business continuity.
🎯 Key Objectives:
- Ensure patient safety
- Maintain product quality
- Achieve regulatory compliance
- Prevent financial and reputational loss
🏗️ Regulatory Framework for Risk Management
🌍 Global Guidelines
Pharmaceutical risk management practices are primarily guided by:
- ICH Q9 – Quality Risk Management
- WHO Technical Report Series (TRS)
- EU GMP Annex 20
- US FDA Guidance on Risk-Based Approaches
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Q9 framework is widely adopted in Bangladesh.
🇧🇩 Bangladesh Regulatory Context
In Bangladesh, risk management is embedded within:
- GMP guidelines enforced by Directorate General of Drug Administration
- Quality Management Systems (QMS) in pharma companies
- Inspection and audit requirements for export markets
Leading companies like Beximco Pharmaceuticals Ltd. and Square Pharmaceuticals Ltd. follow advanced risk-based approaches aligned with global standards.
🔄 Risk Management Lifecycle (ICH Q9 Model)
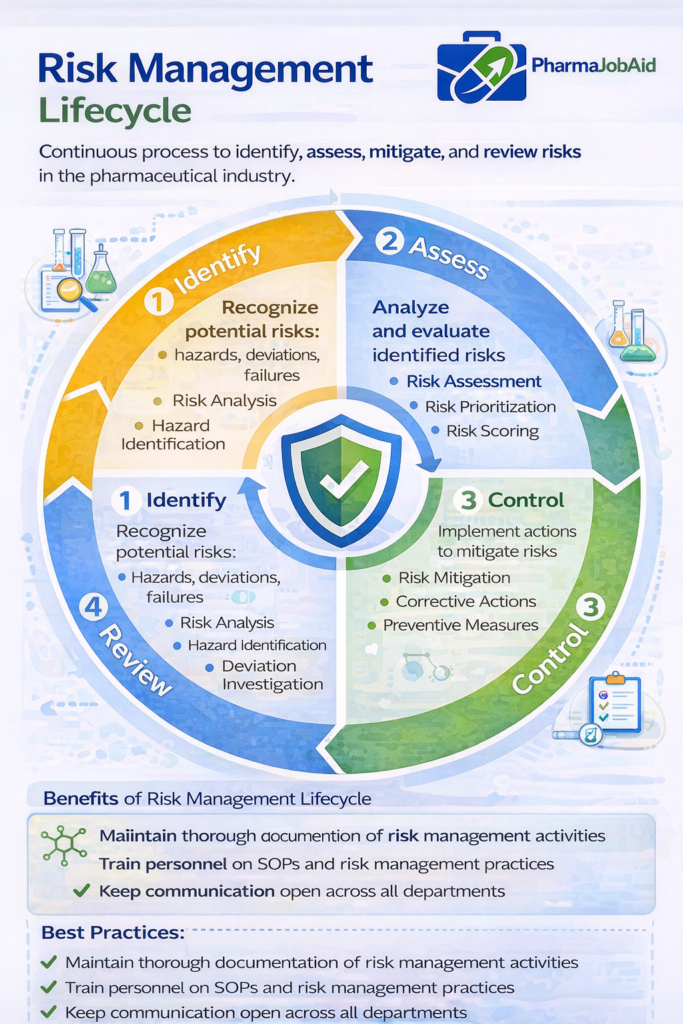
A structured lifecycle ensures consistent risk handling across pharmaceutical operations.
1. Risk Identification
- Process deviations
- Equipment failures
- Contamination risks
- Human error
2. Risk Assessment
- Probability of occurrence
- Severity of impact
- Detectability
3. Risk Control
- Risk reduction (controls, SOPs, automation)
- Risk acceptance (if within limits)
4. Risk Communication
- Cross-functional sharing (QA, QC, Production)
5. Risk Review
- Continuous monitoring
- CAPA integration
🧰 Key Risk Management Tools Used in Pharma

1. Failure Mode and Effects Analysis (FMEA)
🔹 Key Components
- Failure Mode → How the process can fail
- Effect → Impact on product/patient
- Cause → Root cause of failure
- Controls → Existing detection/prevention
📊 Detailed Scoring System
Severity (S)
| Score | Impact |
|---|---|
| 1–3 | Minor (no impact on quality) |
| 4–6 | Moderate (batch deviation) |
| 7–8 | Major (product rejection) |
| 9–10 | Critical (patient risk / recall) |
Occurrence (O)
| Score | Frequency |
|---|---|
| 1–2 | Rare |
| 3–5 | Occasional |
| 6–8 | Frequent |
| 9–10 | Very frequent |
Detectability (D)
| Score | Detection Ability |
|---|---|
| 1–2 | Easily detected |
| 3–5 | Moderate detection |
| 6–8 | Difficult detection |
| 9–10 | Undetectable |
🧮 Formula:
RPN = S × O × D

- Identifies potential failure points
- Calculates Risk Priority Number (RPN)
2. Hazard Analysis and Critical Control Points (HACCP)
7 Core HACCP Principles (Expanded)
1. Hazard Analysis
Identify:
- Biological → microbes
- Chemical → residues
- Physical → particles
2. Determine CCPs
Critical steps where control is essential
Example:
- Sterilization
- Filtration
- Filling
3. Establish Critical Limits
- Temperature (e.g., 121°C sterilization)
- pH
- Time
4. Monitoring System
- Real-time sensors
- Batch records
- QC testing
5. Corrective Actions
If deviation occurs:
- Stop production
- Investigate
- Reject batch if needed
6. Verification
- Audits
- Validation studies
- Microbiological testing
7. Record Keeping
- Batch records
- CCP logs
- Deviation reports
🧪 Pharma HACCP Example
Process: Sterile Injectable
| Step | Hazard | CCP | Control |
|---|---|---|---|
| Filtration | Microbial | Yes | 0.22 micron filter |
| Filling | Contamination | Yes | Laminar airflow |
| Storage | Degradation | No | Temp control |
📊 Types of Hazards
🔬 Biological
- Bacteria, fungi
⚗️ Chemical
- Cross-contamination
⚙️ Physical
- Glass particles
🎯 Benefits in Pharma
- Ensures product safety
- Prevents contamination
- Supports GMP compliance
- Required for export markets
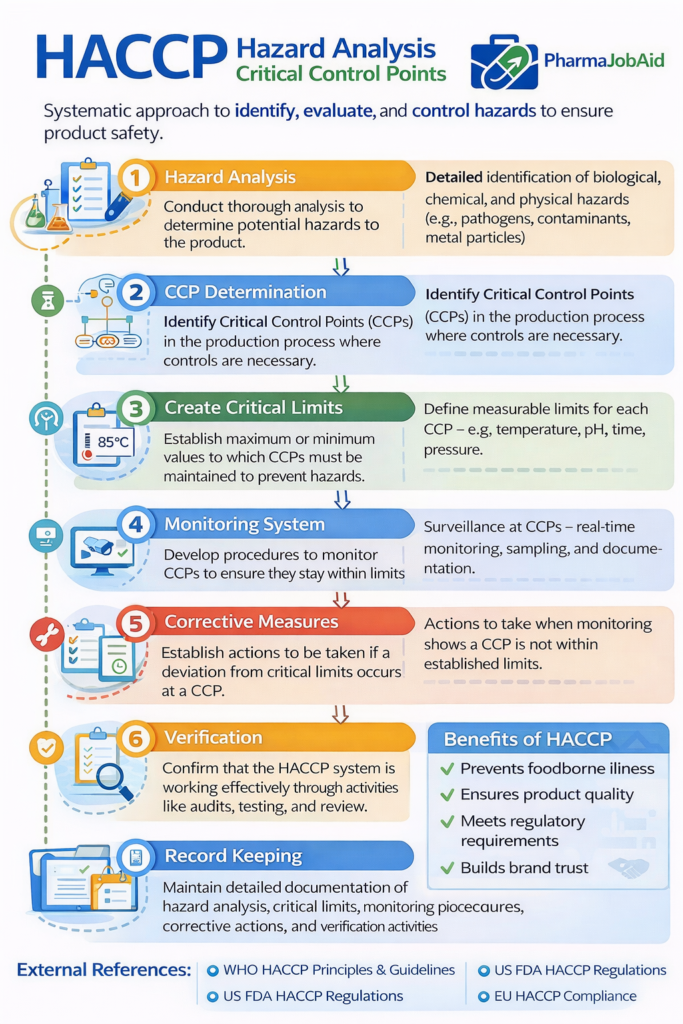
- Focuses on critical process steps
- Widely used in sterile manufacturing
3. Fault Tree Analysis (FTA)
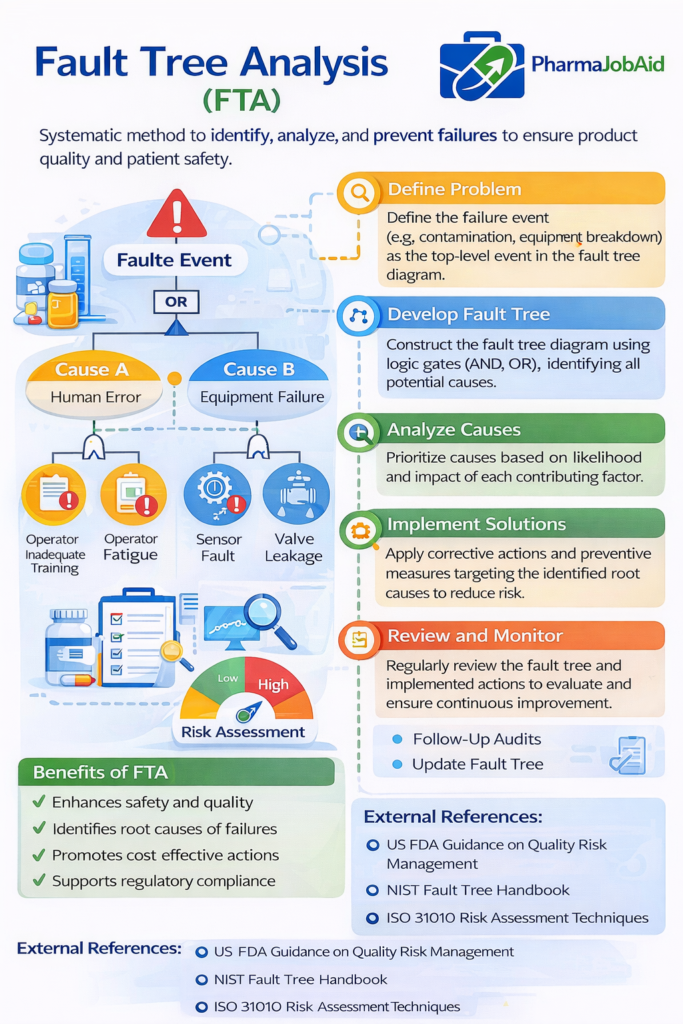
Graphical analysis of system failures
- Useful for root cause investigations
4. Risk Ranking and Filtering (RRF)
- Prioritizes risks based on impact
- Helps allocate resources efficiently
⚠️ Types of Risks in Pharmaceutical Industry
🧪 1. Quality Risks
- Contamination
- OOS (Out of Specification)
- Batch failures
🏭 2. Manufacturing Risks
- Equipment breakdown
- Process variability
- Cross-contamination
📦 3. Supply Chain Risks
- Raw material shortages
- Vendor quality issues
- Logistics disruptions
📜 4. Regulatory Risks
- Non-compliance with GMP
- Audit failures
- Product recalls
💻 5. Data Integrity Risks
- ALCOA+ violations
- Unauthorized data manipulation
🧾 Real-Life Example (Bangladesh Context)
A tablet manufacturing company in Bangladesh faced recurring dissolution failures.
🔎 Investigation:
- Root cause identified: granulation variability
🛠️ Risk Assessment Tool:
- FMEA used to evaluate process parameters
✅ CAPA:
- Process standardization
- Operator retraining
- Equipment calibration
📈 Outcome:
- Batch rejection reduced by 70%
🔗 Integration with Quality Systems
Risk management is not standalone—it integrates with:
- CAPA (Corrective and Preventive Action)
- Deviation Management
- Change Control
- Validation & Qualification
- Self-Inspection / Audits
🤖 Digital Transformation & Risk Management (Pharma 4.0)
Modern pharmaceutical companies are adopting:
- AI-based predictive risk analysis
- Electronic Quality Management Systems (eQMS)
- Real-time monitoring (IoT sensors)
Organizations like International Society for Pharmaceutical Engineering promote risk-based digital transformation under Pharma 4.0.
👨⚕️ Career Opportunities in Risk Management (Bangladesh)
Risk management skills are highly valued in:
🔹 Quality Assurance (QA)
- Risk assessment during deviations
- Audit readiness
🔹 Quality Control (QC)
- Analytical risk evaluation
🔹 Production
- Process risk mitigation
🔹 Regulatory Affairs
- Risk-based submissions
💼 Key Job Roles:
- QA Officer / Executive
- Validation Engineer
- Compliance Specialist
- Risk Management Analyst
🎓 Required Skills:
- Knowledge of ICH Q9
- GMP compliance
- Root cause analysis
- Data integrity principles
📈 Why Risk Management is Critical for Bangladesh Pharma
- Supports export compliance (US FDA, EU GMP)
- Enhances product quality and safety
- Reduces cost of failures
- Builds global trust
Bangladesh aims to become a global pharmaceutical hub—risk management is a key enabler.
🧠 Best Practices for Effective Risk Management
- Adopt risk-based thinking across all departments
- Use standardized tools (FMEA, HACCP)
- Ensure documentation & traceability
- Conduct regular risk reviews
- Integrate with QMS and CAPA
🔗 External References (SEO Backlinks)
- World Health Organization GMP Guidelines
- U.S. Food and Drug Administration Risk-Based Inspection
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Q9 Guideline
🏁 Conclusion
Risk management is the backbone of a robust pharmaceutical quality system. In Bangladesh, as the industry continues to expand globally, adopting structured, science-based risk management practices is essential for sustainability, compliance, and patient safety.
For professionals, mastering risk management tools and frameworks can significantly boost career prospects in QA, QC, and regulatory affairs.
