🔬 Research & Development (R&D) and Product Development (PD) in the Pharmaceutical Industry of Bangladesh
The pharmaceutical industry in Bangladesh has undergone a remarkable transformation over the past three decades. From being heavily dependent on imported medicines, the country has emerged as one of the most self-sufficient pharmaceutical markets in the developing world, meeting approximately 97% of its domestic drug demand.
However, while manufacturing strength has been the backbone of this growth, the next phase of evolution depends on something far more complex and strategic — Research & Development (R&D) and Product Development (PD).
Historically, Bangladesh’s pharmaceutical sector thrived under the TRIPS (Trade-Related Aspects of Intellectual Property Rights) waiver, which allowed local companies to produce patented medicines without facing intellectual property restrictions. This advantage enabled rapid growth in the generic drug segment, making medicines affordable and widely available.
But the global pharmaceutical landscape is changing rapidly.
With Bangladesh expected to graduate from Least Developed Country (LDC) status, the industry will face:
- Stronger patent enforcement
- Increased global competition
- Higher regulatory expectations
- Demand for innovation
This shift means that companies can no longer rely solely on reverse engineering or generic manufacturing. Instead, they must invest in:
- Advanced formulation development
- Complex generics
- Biosimilars
- Novel drug delivery systems
At the center of all these transformations lie R&D and PD functions, which are now considered the core drivers of competitiveness, sustainability, and global expansion.
🏭 2. Bangladesh Pharmaceutical Industry: Detailed Market Analysis
2.1 Market Size and Growth
As of 2026, the pharmaceutical market in Bangladesh is valued at approximately USD 3.5–4 billion, with a consistent annual growth rate of 12–15%.
This makes it one of the fastest-growing industries in the country.
Key Growth Drivers:
- Increasing population
- Rising healthcare awareness
- Expansion of private healthcare facilities
- Growth of middle-class income
- Government healthcare initiatives
2.2 Industry Structure
The pharmaceutical industry in Bangladesh consists of over 250 registered companies, but the market is highly concentrated.
Top Companies:
- Square Pharmaceuticals
- Beximco Pharma
- Incepta Pharmaceuticals
- Renata Limited
- Eskayef Pharmaceuticals
These companies dominate the market and account for a significant portion of total sales.
2.3 Export Market
Bangladesh exports pharmaceutical products to more than 150 countries, including:
- USA
- UK
- EU countries
- Africa
- Southeast Asia
Export growth is driven by:
- WHO GMP compliance
- Competitive pricing
- Increasing regulatory approvals
2.4 R&D Investment Trends
Despite strong growth, R&D investment remains relatively low compared to global standards.
Typical R&D Investment:
- Bangladesh: 1–3% of revenue
- Global pharma: 10–20%
This gap highlights the need for increased focus on innovation.
🔬 3. What is Pharmaceutical R&D?
Pharmaceutical Research & Development (R&D) refers to the scientific process of discovering, designing, and testing new drugs or improving existing ones.
It involves a combination of:
- Chemistry
- Biology
- Pharmacology
- Engineering
- Data analysis
3.1 Types of R&D in Bangladesh
1. Generic R&D
This is the most common form of R&D in Bangladesh.
It involves:
- Reverse engineering of drugs
- Development of equivalent formulations
- Bioequivalence testing
2. Incremental Innovation
This includes improvements such as:
- Extended-release tablets
- Fixed-dose combinations
- Improved stability
3. Advanced R&D (Emerging)
Some leading companies are now exploring:
- Biologics
- Biosimilars
- Oncology drugs
- Vaccines

💊 4. What is Product Development (PD)?
Product Development is the process of transforming a drug concept into a marketable pharmaceutical product.
It involves:
- Formulation design
- Process optimization
- Scale-up
- Manufacturing validation
4.1 Key Objectives of PD
- Ensure product stability
- Achieve desired bioavailability
- Ensure manufacturability
- Meet regulatory requirements
⚖️ 5. R&D vs PD – In-Depth Comparison
| Parameter | R&D | PD |
| Focus | Research & discovery | Product creation |
| Stage | Early | Late |
| Risk | High | Moderate |
| Output | Molecule | Finished product |
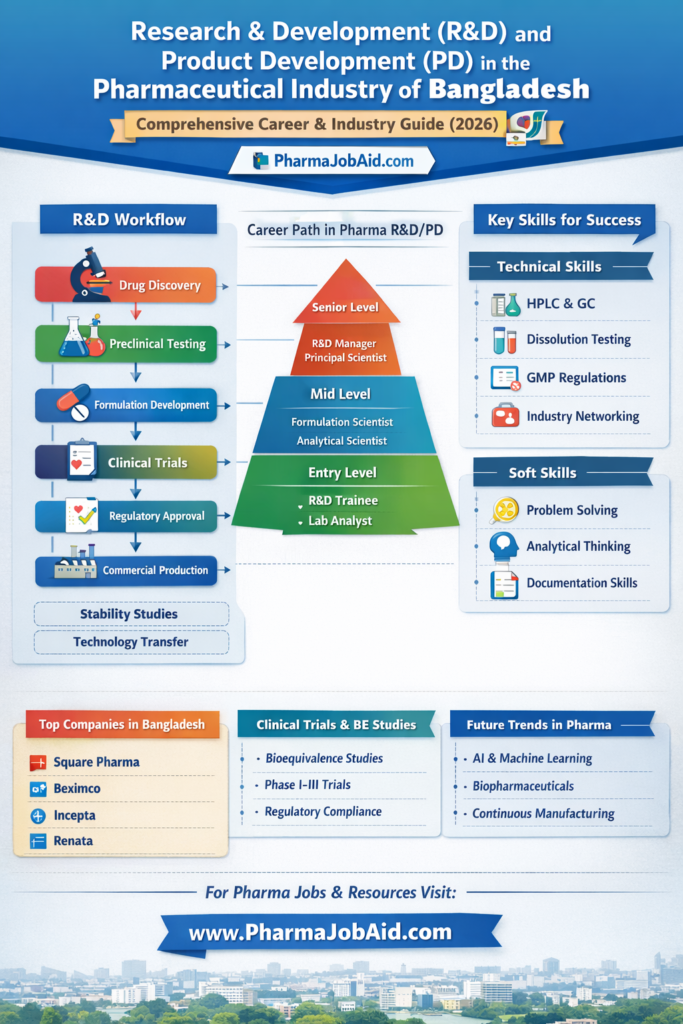
🔁 6. Drug Development Lifecycle (DETAILED)
The drug development lifecycle is a multi-stage process involving scientific, regulatory, and manufacturing activities.
6.1 Stage 1: Drug Selection
In Bangladesh, drug selection is primarily driven by:
- Market demand
- Patent expiration
- Competitive analysis
Companies often select molecules that:
- Have high prescription volume
- Are nearing patent expiry
- Have export potential
6.2 Stage 2: Preformulation Studies
Preformulation studies are critical in understanding the physical and chemical properties of the API (Active Pharmaceutical Ingredient).
Key Parameters:
- Solubility
- Stability
- pKa
- Polymorphism
- Hygroscopicity
These properties directly influence formulation design.
6.3 Stage 3: Formulation Development
Formulation development involves designing a dosage form that delivers the drug effectively.
Types of Dosage Forms:
- Tablets
- Capsules
- Syrups
- Injectables
6.4 Stage 4: Analytical Development
Analytical development ensures product quality and consistency.
Key Techniques:
- HPLC
- GC
- UV spectroscopy
6.5 Stage 5: Bioequivalence (BE)
BE studies ensure that the generic product performs similarly to the reference drug.
6.6 Stage 6: Technology Transfer
This involves transferring the product from R&D to manufacturing.
6.7 Stage 7: Commercial Production
Final stage where the product is manufactured at scale.
🧪 7. Formulation Development (FULL DEEP DIVE)
Formulation development is one of the most critical steps in pharmaceutical product development.
7.1 Tablet Formulation Techniques
1. Wet Granulation
- Improves flowability
- Enhances compressibility
2. Direct Compression
- Cost-effective
- Faster process
3. Dry Granulation
- Suitable for moisture-sensitive drugs
7.2 Challenges in Formulation
- Poor solubility
- Stability issues
- Dissolution variability
📋 SOP: Tablet Formulation (FULL VERSION)
Objective:
To develop a stable and bioequivalent tablet formulation.
Step 1: API Characterization
- Determine solubility
- Measure particle size
Step 2: Excipient Selection
- Binder
- Disintegrant
- Lubricant
Step 3: Trial Batch Preparation
- Prepare multiple batches
Step 4: Evaluation
- Hardness
- Friability
- Dissolution
Step 5: Optimization
- Use Design of Experiments (DoE)
🧫 8. Analytical Development (DEEP VERSION)
Analytical development ensures the drug product meets quality standards.
8.1 Key Analytical Tests
- Assay
- Dissolution
- Impurity profiling
📋 SOP: HPLC Method Development
Step 1: Column Selection
Step 2: Mobile Phase Optimization
Step 3: Detection Wavelength Selection
🧬 9. Bioequivalence (BE) Studies — Complete Scientific & Regulatory Guide
Bioequivalence (BE) is the most critical requirement for generic drug approval in Bangladesh and globally. Without BE data, a generic drug cannot demonstrate that it performs the same as the innovator product.
9.1 What is Bioequivalence?
Bioequivalence means that two pharmaceutical products (test and reference) show:
- Comparable bioavailability
- Similar rate and extent of absorption
In simpler terms:
👉 The generic drug must work exactly like the original brand drug.
9.2 Why BE Studies Are Critical in Bangladesh
Since Bangladesh is largely a generic drug market, BE studies ensure:
- Therapeutic equivalence
- Patient safety
- Regulatory compliance
- Export approval
Countries like the USA, EU, and WHO prequalification programs require BE data.
9.3 Types of Bioequivalence Studies
1. In Vivo BE Studies
- Conducted in human volunteers
- Measures drug concentration in plasma
2. In Vitro BE Studies
- Dissolution-based
- Used for BCS Class I drugs
9.4 Study Design (DETAILED)
Standard Design:
- Randomized
- Two-period crossover
- Two-sequence
Example:
- 24 healthy volunteers
- Single-dose study
9.5 Key Pharmacokinetic Parameters
| Parameter | Meaning |
| Cmax | Peak plasma concentration |
| Tmax | Time to reach Cmax |
| AUC | Total drug exposure |
9.6 Sample BE Data (REALISTIC EXPANDED)
| Parameter | Test Product | Reference Product |
| Cmax | 120 ng/mL | 118 ng/mL |
| Tmax | 2.1 hr | 2.0 hr |
| AUC0-t | 980 ng·hr/mL | 960 ng·hr/mL |
| AUC0-∞ | 1050 ng·hr/mL | 1035 ng·hr/mL |
9.7 Acceptance Criteria
According to regulatory guidelines:
👉 90% Confidence Interval must fall within:
80% – 125%
9.8 BE Study Workflow (STEP-BY-STEP SOP)
Step 1: Protocol Development
- Study design
- Inclusion/exclusion criteria
Step 2: Ethics Approval
- Institutional Review Board (IRB)
Step 3: Volunteer Recruitment
- Healthy subjects
Step 4: Dosing
- Controlled administration
Step 5: Sample Collection
- Blood sampling at specific intervals
Step 6: Bioanalysis
- LC-MS/MS analysis
Step 7: Data Analysis
- Pharmacokinetic calculations
9.9 BE Challenges in Bangladesh
- Limited BE centers
- High study cost
- Regulatory delays
🧪 10. Stability Studies — Full Regulatory & Industrial Perspective
Stability studies determine how the quality of a drug product changes over time.
10.1 Purpose of Stability Studies
- Determine shelf life
- Ensure product safety
- Validate packaging
10.2 Types of Stability Studies
1. Long-Term Stability
- 25°C / 60% RH
2. Accelerated Stability
- 40°C / 75% RH
3. Intermediate Stability
- 30°C / 65% RH
10.3 Stability Study Design
| Time Point | Testing |
| 0 month | Initial |
| 3 months | Accelerated |
| 6 months | Accelerated |
| 12 months | Long-term |
📋 SOP: Stability Study (FULL INDUSTRIAL VERSION)
Objective:
To evaluate product stability under defined environmental conditions.
Step 1: Batch Selection
- Minimum 3 batches
- Pilot or production scale
Step 2: Packaging
- Final market packaging
Step 3: Storage Conditions
- Controlled chambers
Step 4: Testing Parameters
- Assay
- Dissolution
- Degradation products
Step 5: Data Evaluation
- Trend analysis
- Shelf-life estimation
10.4 Stability Challenges in Bangladesh
- Climatic zone IV (hot & humid)
- Packaging sensitivity
- Storage logistics
🏭 11. Technology Transfer (TT) — Complete Industrial SOP
Technology Transfer is the process of transferring knowledge from R&D to manufacturing.
11.1 Types of Technology Transfer
1. Internal TT
- R&D → Production
2. External TT
- Company → Contract manufacturer
11.2 Technology Transfer Workflow
- Process documentation
- Pilot batch
- Validation batch
- Commercial batch
📋 SOP: Technology Transfer
Step 1: TT Protocol Preparation
- Product details
- Process parameters
Step 2: Risk Assessment
- Identify critical parameters
Step 3: Trial Batch Manufacturing
- Pilot-scale production
Step 4: Process Validation
- Consistency check
Step 5: TT Report
- Final documentation
11.3 Key Challenges
- Scale mismatch
- Equipment differences
- Process reproducibility
📈 12. Scale-Up & Process Optimization
Scaling up from lab to production is one of the most complex stages.
12.1 Key Objectives
- Maintain product quality
- Ensure batch consistency
- Reduce cost
12.2 Common Scale-Up Issues
- Mixing uniformity
- Granulation variability
- Dissolution failure
12.3 Optimization Techniques
- Design of Experiments (DoE)
- Process Analytical Technology (PAT)
📋 13. Regulatory Framework — Bangladesh & Global
Regulatory compliance is essential for product approval and export.
13.1 Bangladesh Regulatory Authority
- Directorate General of Drug Administration (DGDA)
13.2 Global Regulatory Bodies
- US FDA
- EMA
- WHO
13.3 Key Guidelines
- ICH Q8 – Pharmaceutical Development
- ICH Q9 – Risk Management
- ICH Q10 – Quality System
13.4 Common Regulatory Requirements
- Dossier submission
- Stability data
- BE study data
🧬 14. Case Study 1: Beximco Pharma (COVID-19 Response)
Beximco Pharma demonstrated rapid R&D capability during the COVID-19 pandemic.
Key Achievements:
- Developed generic Remdesivir
- Exported globally
- Accelerated regulatory approval
Lessons:
- Strong R&D infrastructure enables rapid response
- Regulatory agility is critical
💊 15. Case Study 2: Square Pharmaceuticals
Square is the largest pharmaceutical company in Bangladesh.
R&D Strategy:
- Focus on generics
- Continuous product pipeline
- Export-oriented development
Success Factors:
- Strong formulation team
- High-quality manufacturing
💉 16. Case Study 3: Beacon Pharmaceuticals (Oncology)
Beacon Pharma specializes in oncology drugs.
Key Strengths:
- First mover in cancer drugs
- Advanced formulation capability
Impact:
- Export growth
- High-value product segment
🧬 17. Emerging R&D Areas in Bangladesh
1. Biologics
- Vaccines
- Monoclonal antibodies
2. Nanotechnology
- Targeted drug delivery
3. AI & Machine Learning
- Drug discovery
- Predictive modeling
⚠️ 18. Challenges in Pharma R&D (Bangladesh Context)
- Low R&D investment
- Lack of innovation culture
- Skilled manpower shortage
🚀 19. Future Outlook
Bangladesh pharma is transitioning toward:
- Complex generics
- Biosimilars
- Global exports
👨🔬 20. Career Path in R&D and Product Development (Bangladesh)
The R&D and Product Development sector offers one of the most intellectually rewarding and financially stable career paths in the pharmaceutical industry.
Unlike sales or production roles, R&D careers are:
- Knowledge-intensive
- Research-driven
- Globally transferable
20.1 Entry-Level Career Opportunities
Fresh graduates in pharmacy, chemistry, or related fields typically start in:
1. R&D Trainee / Executive
- Assists in formulation trials
- Supports documentation
- Conducts basic experiments
2. Analytical Chemist
- Performs HPLC analysis
- Conducts dissolution testing
- Supports method validation
3. Quality Control (QC) Support (Transition Role)
- Many professionals shift from QC → R&D
20.2 Mid-Level Roles
After 3–5 years of experience:
1. Formulation Scientist
- Designs drug formulations
- Optimizes dissolution profiles
2. Analytical Scientist
- Develops and validates methods
- Handles impurity profiling
3. Technology Transfer Specialist
- Bridges R&D and production
20.3 Senior-Level Roles
After 8–15 years:
1. R&D Manager
- Leads projects
- Oversees development pipeline
2. Head of Product Development
- Strategic decision-making
- Portfolio planning
3. Director of R&D
- Global strategy
- Innovation leadership
💰 21. Salary Structure in Bangladesh (REALISTIC MARKET DATA)
Entry-Level (0–2 years)
- BDT 25,000 – 40,000
Mid-Level (3–7 years)
- BDT 50,000 – 90,000
Senior-Level (8+ years)
- BDT 100,000 – 250,000+
21.1 Factors Affecting Salary
- Company size (e.g., Square vs small firms)
- Skill specialization
- Experience
- Regulatory exposure
🧠 22. Skills Required for R&D Careers
22.1 Technical Skills
Essential:
- HPLC operation
- Dissolution testing
- Stability study knowledge
- GMP compliance
Advanced Skills:
- Method validation
- DoE (Design of Experiments)
- Regulatory documentation
22.2 Soft Skills
- Problem-solving
- Analytical thinking
- Documentation skills
- Attention to detail
🧰 23. Tools & Technologies Used in R&D
Analytical Tools:
- HPLC
- GC
- UV Spectrophotometer
Software:
- Empower (HPLC software)
- LIMS
- Statistical tools
🎯 24. How to Get a Job in R&D (STEP-BY-STEP)
Step 1: Academic Qualification
- B.Pharm / M.Pharm
Step 2: Internship
- Pharma company training
Step 3: Skill Development
- Learn HPLC
- Learn GMP
Step 4: Apply Smartly
👉 Use: https://pharmajobaid.com/
24.1 Resume Tips
- Highlight lab experience
- Mention instruments
- Include projects
24.2 Interview Preparation
Common questions:
- What is dissolution?
- What is BE study?
- Explain HPLC principle
🌐 26. External Linking Strategy (Authority SEO)
Linking to high-authority websites increases trust.
Recommended External Links:
- WHO (GMP guidelines)
- US FDA
- EMA
- ICH
🚀 31. Monetization Strategy (BONUS)
PharmaJobAid can generate revenue through:
- Job postings
- Resume services
- Training programs
🏁 32. FINAL CONCLUSION
R&D and Product Development are the future of Bangladesh’s pharmaceutical industry.
As the country transitions from a generic-driven market to an innovation-driven ecosystem, the demand for skilled professionals in R&D will continue to grow.
For students, professionals, and job seekers, this field offers:
✔ High career growth
✔ Global opportunities
✔ Long-term stability
Top of Form
Bottom of Form
