Internal Audits in the Pharmaceutical Industry in Bangladesh: A Complete GMP Compliance Guide for Pharma Professionals
📌 Introduction
Internal audits are a cornerstone of pharmaceutical quality systems, ensuring that companies consistently meet regulatory requirements, maintain product quality, and operate in compliance with Good Manufacturing Practices (GMP). In Bangladesh, where the pharmaceutical sector is rapidly growing and exporting to global markets, internal audits play a vital role in maintaining international standards.
Regulatory bodies such as the Directorate General of Drug Administration (DGDA), along with global guidelines from the World Health Organization and U.S. Food and Drug Administration, emphasize the importance of internal audits as part of a robust Quality Management System (QMS).
🔍 What is an Internal Audit in Pharma?
An internal audit is a systematic, independent, and documented process for obtaining evidence and evaluating it objectively to determine the extent to which audit criteria are fulfilled.
In pharmaceutical companies, internal audits assess:
- GMP compliance
- SOP adherence
- Data integrity (ALCOA+)
- Regulatory readiness
- Process effectiveness
🎯 Objectives of Internal Audits
Internal audits are not just about finding faults—they are tools for continuous improvement.
Key Objectives:
- Ensure compliance with GMP and regulatory guidelines
- Identify gaps and risks in processes
- Improve operational efficiency
- Verify implementation of CAPA (Corrective and Preventive Actions)
- Prepare for external inspections (DGDA, WHO, US FDA)
- Strengthen data integrity practices
🏭 Scope of Internal Audits in Pharma
Internal audits cover all critical departments within a pharmaceutical organization:
1. Quality Assurance (QA)
- SOP compliance
- Batch record review
- Change control
- Deviation management
2. Quality Control (QC)
- Analytical testing
- Laboratory practices
- Instrument calibration
- Stability studies
3. Production
- Manufacturing processes
- Line clearance
- Equipment cleaning validation
- Documentation practices
4. Warehouse & Supply Chain
- Material handling
- Storage conditions
- Inventory control
- Distribution practices
5. Engineering & Maintenance
- HVAC systems
- Preventive maintenance
- Utility systems (water, steam, compressed air)
6. Regulatory Affairs
- Dossier management
- Compliance with international guidelines
🔄 Internal Audit Lifecycle
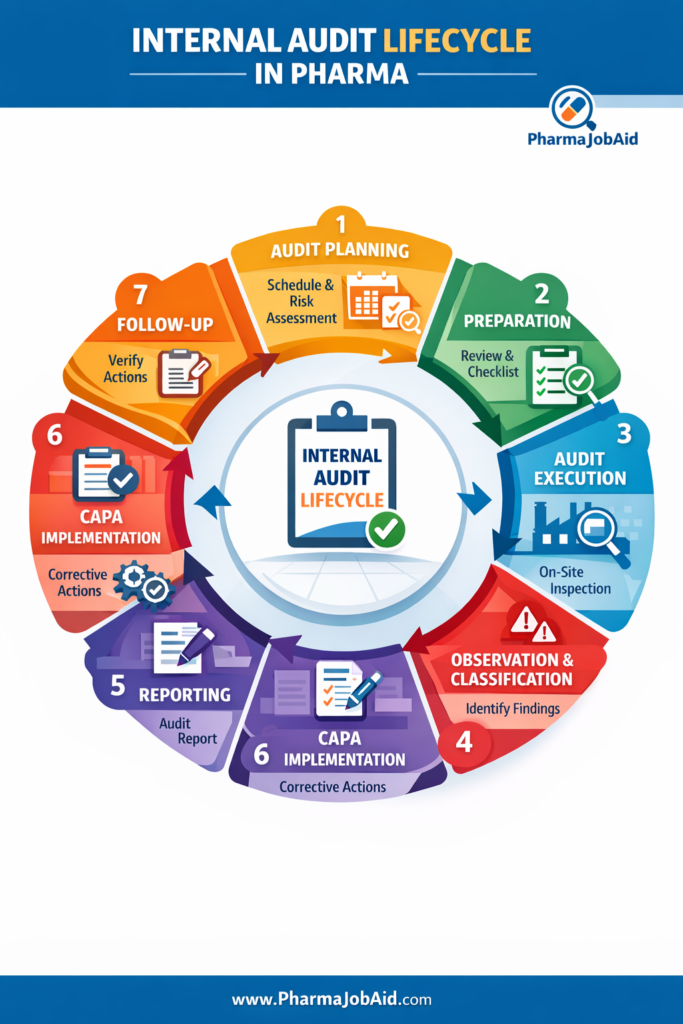
Step-by-Step Process:
1. Audit Planning
- Annual audit schedule prepared by QA
- Risk-based prioritization
- Selection of trained auditors
2. Preparation
- Review SOPs, previous audit reports
- Prepare audit checklist
- Define audit scope and objectives
3. Audit Execution
- Opening meeting
- On-site inspection
- Interviews with personnel
- Document review
4. Observation & Classification
Findings are categorized as:
- Critical
- Major
- Minor
5. Reporting
- Audit report issued within defined timeline
- Clear documentation of findings
6. CAPA Implementation
- Root cause analysis
- Corrective actions
- Preventive actions
7. Follow-Up
- Verification of CAPA effectiveness
- Closure of audit findings
📊 Types of Internal Audits in Pharma
🔹 System Audit
Evaluates the overall Quality Management System (QMS)
🔹 Process Audit
Focuses on specific processes like manufacturing or testing
🔹 Product Audit
Examines finished product compliance and batch records
🔹 Compliance Audit
Ensures adherence to regulatory standards (GMP, WHO, FDA)
⚠️ Common Audit Findings in Bangladesh Pharma Industry
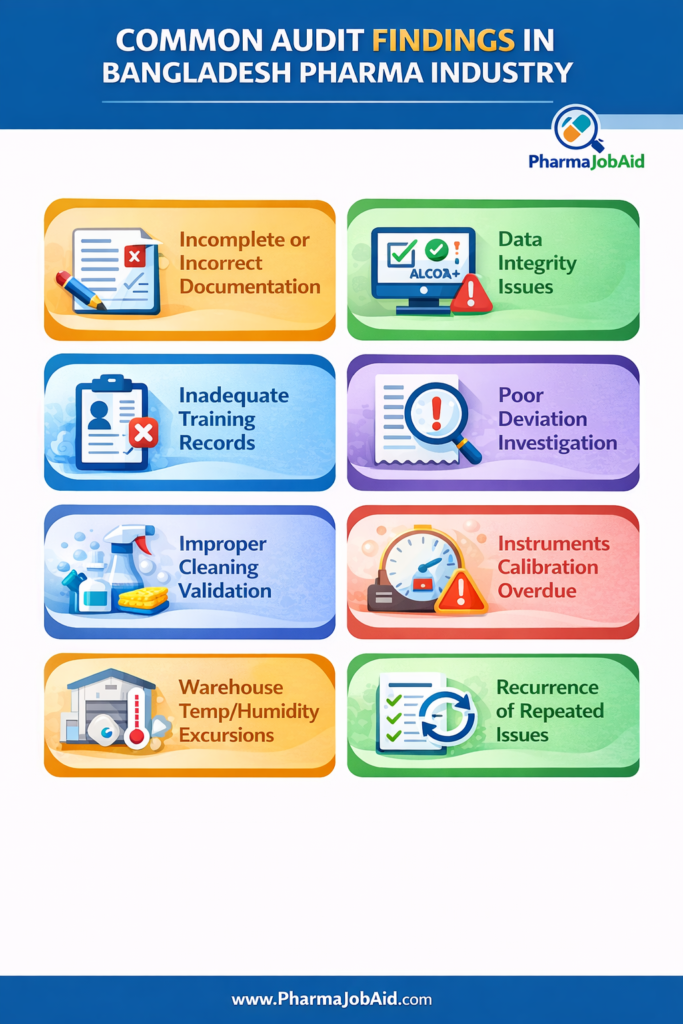
Frequently Observed Issues:
- Incomplete or incorrect documentation
- Data integrity violations (missing signatures, overwriting)
- Inadequate training records
- Poor deviation investigation
- Improper cleaning validation
- Calibration overdue for instruments
- Warehouse temperature/humidity excursions
🧪 Regulatory Expectations in Bangladesh
Pharmaceutical companies in Bangladesh must comply with:
- Directorate General of Drug Administration guidelines
- World Health Organization
- International Council for Harmonisation
- U.S. Food and Drug Administration (for export markets)
Internal audits are mandatory to demonstrate continuous compliance and inspection readiness.
🧩 Role of Internal Audit in QMS
Internal audits are an integral part of a pharmaceutical Quality Management System:
- Linked with CAPA system
- Supports change control
- Strengthens risk management
- Ensures continuous improvement
They act as a self-inspection mechanism, as required by WHO GMP.
📈 Benefits of Internal Audits
✔ For Organizations:
- Improved compliance
- Reduced regulatory risk
- Enhanced product quality
- Better inspection outcomes
✔ For Professionals:
- Strong understanding of GMP
- Career growth in QA/QC
- Expertise in regulatory compliance
🛠️ Best Practices for Effective Internal Audits
- Use risk-based audit planning
- Ensure auditor independence
- Maintain detailed audit checklists
- Focus on data integrity (ALCOA+)
- Conduct regular auditor training
- Implement digital audit tools
- Track CAPA effectiveness
📌 Practical Example (Bangladesh Pharma Context)
Scenario:
A tablet manufacturing facility in Bangladesh undergoes an internal audit.
Findings:
- Batch Manufacturing Record (BMR) missing operator signature
- Temperature log gaps in warehouse
- Cleaning validation not updated
Actions Taken:
- CAPA initiated
- Retraining of staff
- SOP revision
- Increased audit frequency
Outcome:
- Improved compliance
- Successful DGDA inspection
📊 Internal Audit Checklist (Sample)
- SOP availability and compliance
- Training records updated
- Equipment calibration status
- Environmental monitoring data
- Batch documentation completeness
- Deviation and CAPA records
🚀 Future Trends in Internal Auditing
- Digital audit systems (eQMS)
- AI-driven compliance monitoring
- Remote audits (post-COVID era)
- Data integrity automation tools
🧾 Conclusion
Internal audits are the backbone of pharmaceutical compliance in Bangladesh. As the industry expands into regulated markets, maintaining high standards through robust internal auditing systems is no longer optional—it is essential.
Companies that invest in structured internal audit programs not only ensure regulatory compliance but also gain a competitive edge in global markets.
