🧪 Management Review Meeting (MRM) in the Pharmaceutical Industry: GMP-Compliant Guide with WHO, ICH Q10 & DGDA Regulatory References
📌 Introduction
In the pharmaceutical industry, quality is not just a goal—it is a regulatory obligation. One of the most critical mechanisms to ensure quality system effectiveness is the Management Review Meeting (MRM).
In Bangladesh, pharmaceutical companies must comply with regulations set by the Directorate General of Drug Administration (DGDA) while also aligning with international frameworks such as the World Health Organisation GMP and International Council for Harmonization.
MRM acts as a bridge between operational performance and top management oversight, ensuring that the Pharmaceutical Quality System (PQS) remains effective, compliant, and continuously improving.
🔍 Definition of MRM (Regulatory Perspective)
📖 According to International Council for Harmonisation:
“Senior management has the ultimate responsibility to ensure an effective pharmaceutical quality system is in place and is periodically reviewed for suitability and effectiveness.”
👉 This periodic review is executed through Management Review Meetings (MRM).
📚 Regulatory Guideline References for MRM
🌍 1. WHO GMP (TRS Guidelines)
World Health Organization GMP (e.g., TRS 986, TRS 1025)
🔹 Key Requirements:
- Senior management must ensure:
- Quality system effectiveness
- Continuous improvement
- Product quality consistency
🔹 Relevant Clauses:
- WHO GMP Chapter 1 (Quality Assurance)
- Requires:
- Regular review of:
- Product quality
- Process performance
- Deviations & CAPA
- Regular review of:
📌 WHO emphasizes:
“Management should review the quality system at regular intervals.”
📘 2. ICH Q10 – Pharmaceutical Quality System
International Council for Harmonisation
🔹 Section 2.6 – Management Review of Process Performance & Product Quality
ICH Q10 defines two key management review elements:
✅ a) Management Review of Process Performance
Includes:
- Process capability
- Deviations
- CAPA effectiveness
✅ b) Management Review of Product Quality
Includes:
- APQR/PQR trends
- Stability data
- Complaints & recalls
📌 Key Concept:
MRM ensures knowledge management + quality risk management integration
🇧🇩 3. DGDA Bangladesh Requirements
Directorate General of Drug Administration
While DGDA follows WHO GMP, inspectors typically expect:
- Documented MRM procedure (SOP)
- Defined frequency (quarterly/biannual)
- Evidence of:
- CAPA follow-up
- Audit review
- Trend analysis
- Signed meeting minutes
📌 During DGDA audits, lack of effective MRM may result in:
- Major observation
- Weak QMS rating
🎯 Objectives of MRM (Guideline-Aligned)
Based on WHO + ICH Q10:
- Ensure PQS effectiveness
- Monitor process performance
- Review product quality
- Identify risks (ICH Q9 integration)
- Ensure regulatory compliance
- Drive continual improvement
🔄 MRM Lifecycle (GMP-Compliant)
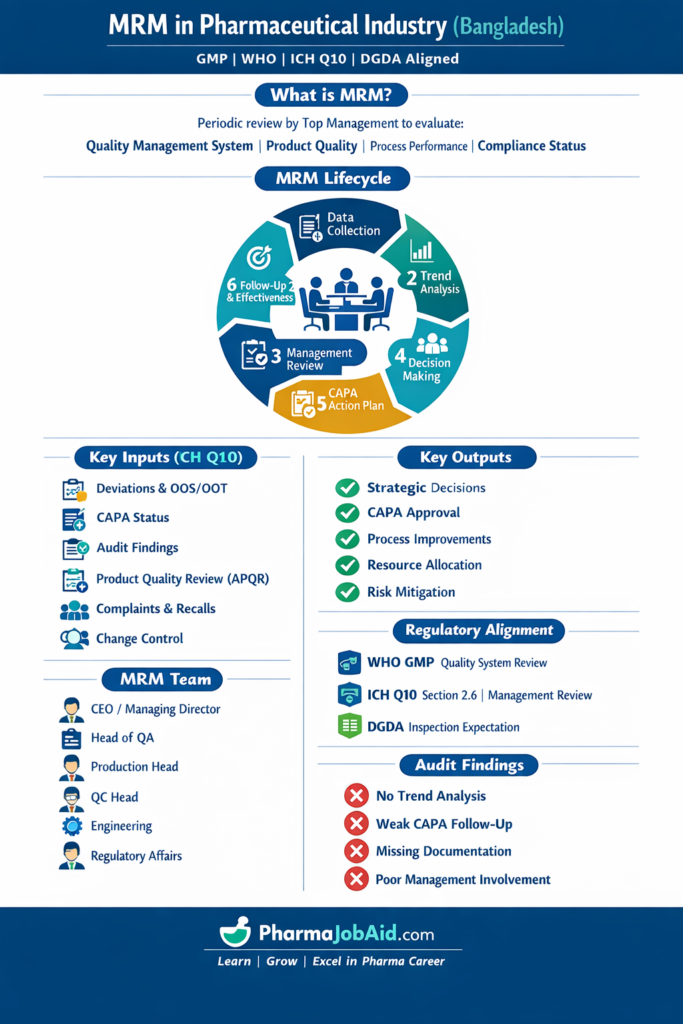
Step-by-Step (Aligned with ICH Q10):
- Data Collection (ICH Q10 Inputs)
- Deviations
- OOS/OOT
- CAPA
- Complaints
- Audit findings
- Data Analysis (Risk-Based)
- Trend analysis
- Risk identification (ICH Q9)
- Management Review Meeting
- Evaluate PQS effectiveness
- Identify gaps
- Decision Making
- Resource allocation
- Process improvements
- Action Plan (CAPA)
- Assign responsibility
- Define timelines
- Follow-up
- Verify effectiveness
📊 MRM Inputs (ICH Q10 Section 2.6)
📌 Mandatory Inputs:
🔹 Product Quality Review (APQR/PQR)
- Batch trends
- Stability data
🔹 Process Performance
- Yield
- Process deviations
🔹 CAPA System
- Open vs closed CAPA
- Effectiveness checks
🔹 Change Control
- Impact of changes
🔹 Audit Results
- Internal & external audits
🔹 Complaints & Recalls
- Market feedback
📈 MRM Outputs (Regulatory Expectation)
According to WHO & ICH:
- Improvement actions
- Resource needs
- CAPA decisions
- Policy updates
- Risk mitigation plans
📌 Outputs must be:
- Documented
- Approved by top management
- Tracked for effectiveness
👥 Roles & Responsibilities (ICH Q10 Alignment)
🔹 Senior Management:
- Ensure PQS effectiveness
- Provide resources
- Review outcomes
🔹 Quality Assurance (QA):
- Organize MRM
- Present data
- Track CAPA
🔹 Department Heads:
- Provide input data
- Implement actions
📅 Frequency of MRM (Guideline Insight)
- WHO: “Periodic” (not fixed)
- Industry Practice:
- Quarterly (recommended)
- Bi-annual (minimum)
📌 High-risk facilities → more frequent reviews
⚠️ Common Regulatory Findings (Bangladesh Context)
DGDA & international audits often identify:
- No trend analysis
- Incomplete CAPA follow-up
- Missing MRM minutes
- Lack of management involvement
💡 Best Practices (Guideline-Based)
✅ Follow ICH Q10 Structure
Align MRM with PQS lifecycle.
✅ Integrate ICH Q9 (Risk Management)
Use risk-based decision-making.
✅ Use Data Integrity Principles (ALCOA+)
- Attributable
- Legible
- Contemporaneous
- Original
- Accurate
✅ Maintain Documentation (WHO GMP)
- Signed minutes
- Action tracking log
🧾 Sample MRM Agenda (Regulatory-Compliant)
- Opening by Management
- Previous Action Review
- PQS Performance Review (ICH Q10)
- Product Quality Review (APQR)
- Audit & Inspection Findings
- CAPA Effectiveness
- Risk Assessment Updates
- Resource Evaluation
- Regulatory Updates
- Action Plan Approval
🧪 Real Audit Scenario (Bangladesh)
During a DGDA inspection:
Observation:
- MRM conducted but no trend analysis
Regulatory Concern:
- Weak PQS effectiveness
Corrective Action:
- Introduced KPI dashboard
- Implemented quarterly trend analysis
🚀 Career Relevance of MRM
Understanding MRM is essential for:
- QA Officers
- Regulatory Affairs Professionals
- Production Pharmacists
📌 Required Skills:
- GMP knowledge (WHO, ICH)
- Data analysis
- Risk assessment
- Documentation
🔗 External Regulatory References (SEO Boost)
- WHO GMP TRS 986 / 1025
- ICH Q10 Pharmaceutical Quality System
- ICH Q9 Quality Risk Management
- DGDA Bangladesh Guidelines
📌 Conclusion
Management Review Meeting (MRM) is a regulatory backbone of the Pharmaceutical Quality System.
Aligned with WHO GMP, ICH Q10, and DGDA expectations, MRM ensures:
- Sustainable compliance
- Continuous improvement
- Product quality assurance
For Bangladesh’s growing pharmaceutical sector, effective MRM is not just compliance—it is a competitive advantage.
